Filter
Associated Lab
- Ahrens Lab (2) Apply Ahrens Lab filter
- Aso Lab (3) Apply Aso Lab filter
- Baker Lab (1) Apply Baker Lab filter
- Betzig Lab (8) Apply Betzig Lab filter
- Bock Lab (1) Apply Bock Lab filter
- Branson Lab (7) Apply Branson Lab filter
- Card Lab (4) Apply Card Lab filter
- Cardona Lab (8) Apply Cardona Lab filter
- Cui Lab (1) Apply Cui Lab filter
- Dickson Lab (1) Apply Dickson Lab filter
- Druckmann Lab (3) Apply Druckmann Lab filter
- Dudman Lab (4) Apply Dudman Lab filter
- Eddy/Rivas Lab (1) Apply Eddy/Rivas Lab filter
- Feliciano Lab (1) Apply Feliciano Lab filter
- Fetter Lab (4) Apply Fetter Lab filter
- Funke Lab (1) Apply Funke Lab filter
- Gonen Lab (11) Apply Gonen Lab filter
- Grigorieff Lab (6) Apply Grigorieff Lab filter
- Harris Lab (5) Apply Harris Lab filter
- Heberlein Lab (1) Apply Heberlein Lab filter
- Hermundstad Lab (1) Apply Hermundstad Lab filter
- Hess Lab (4) Apply Hess Lab filter
- Jayaraman Lab (4) Apply Jayaraman Lab filter
- Ji Lab (5) Apply Ji Lab filter
- Keleman Lab (1) Apply Keleman Lab filter
- Keller Lab (2) Apply Keller Lab filter
- Lavis Lab (16) Apply Lavis Lab filter
- Lee (Albert) Lab (6) Apply Lee (Albert) Lab filter
- Leonardo Lab (2) Apply Leonardo Lab filter
- Lippincott-Schwartz Lab (9) Apply Lippincott-Schwartz Lab filter
- Liu (Zhe) Lab (5) Apply Liu (Zhe) Lab filter
- Looger Lab (6) Apply Looger Lab filter
- Magee Lab (2) Apply Magee Lab filter
- Menon Lab (1) Apply Menon Lab filter
- Pachitariu Lab (1) Apply Pachitariu Lab filter
- Reiser Lab (6) Apply Reiser Lab filter
- Riddiford Lab (1) Apply Riddiford Lab filter
- Romani Lab (6) Apply Romani Lab filter
- Rubin Lab (15) Apply Rubin Lab filter
- Saalfeld Lab (4) Apply Saalfeld Lab filter
- Scheffer Lab (4) Apply Scheffer Lab filter
- Schreiter Lab (4) Apply Schreiter Lab filter
- Shroff Lab (1) Apply Shroff Lab filter
- Simpson Lab (2) Apply Simpson Lab filter
- Singer Lab (6) Apply Singer Lab filter
- Spruston Lab (1) Apply Spruston Lab filter
- Stern Lab (8) Apply Stern Lab filter
- Sternson Lab (2) Apply Sternson Lab filter
- Svoboda Lab (9) Apply Svoboda Lab filter
- Truman Lab (6) Apply Truman Lab filter
- Turaga Lab (3) Apply Turaga Lab filter
- Turner Lab (2) Apply Turner Lab filter
- Wu Lab (1) Apply Wu Lab filter
- Zlatic Lab (7) Apply Zlatic Lab filter
Associated Project Team
- Fly Descending Interneuron (1) Apply Fly Descending Interneuron filter
- Fly Functional Connectome (4) Apply Fly Functional Connectome filter
- Fly Olympiad (1) Apply Fly Olympiad filter
- FlyEM (4) Apply FlyEM filter
- FlyLight (2) Apply FlyLight filter
- GENIE (3) Apply GENIE filter
- ThalamoSeq (1) Apply ThalamoSeq filter
- Tool Translation Team (T3) (3) Apply Tool Translation Team (T3) filter
- Transcription Imaging (6) Apply Transcription Imaging filter
Associated Support Team
- Anatomy and Histology (2) Apply Anatomy and Histology filter
- Cryo-Electron Microscopy (4) Apply Cryo-Electron Microscopy filter
- Electron Microscopy (1) Apply Electron Microscopy filter
- Fly Facility (1) Apply Fly Facility filter
- Integrative Imaging (1) Apply Integrative Imaging filter
- Project Technical Resources (1) Apply Project Technical Resources filter
- Quantitative Genomics (2) Apply Quantitative Genomics filter
- Scientific Computing Software (9) Apply Scientific Computing Software filter
- Viral Tools (1) Apply Viral Tools filter
- Vivarium (1) Apply Vivarium filter
Publication Date
- December 2017 (15) Apply December 2017 filter
- November 2017 (11) Apply November 2017 filter
- October 2017 (7) Apply October 2017 filter
- September 2017 (14) Apply September 2017 filter
- August 2017 (15) Apply August 2017 filter
- July 2017 (20) Apply July 2017 filter
- June 2017 (18) Apply June 2017 filter
- May 2017 (25) Apply May 2017 filter
- April 2017 (21) Apply April 2017 filter
- March 2017 (15) Apply March 2017 filter
- February 2017 (7) Apply February 2017 filter
- January 2017 (18) Apply January 2017 filter
- Remove 2017 filter 2017
186 Janelia Publications
Showing 101-110 of 186 resultsSeconds-scale network states, affecting many neurons within a network, modulate neural activity by complementing fast integration of neuron-specific inputs that arrive in the milliseconds before spiking. Non-rhythmic subthreshold dynamics at intermediate timescales, however, are less well-characterized. We found, using automated whole cell patch clamping in vivo, that spikes recorded in CA1 and barrel cortex in awake mice are often preceded not only by monotonic voltage rises lasting milliseconds, but also by more gradual (lasting 10s-100s of ms) depolarizations. The latter exert a gating function on spiking, in a fashion that depends on the gradual rise duration: the probability of spiking was higher for longer gradual rises, even controlling for the amplitude of the gradual rises. Barrel cortex double-autopatch recordings show that gradual rises are shared across some but not all neurons. The gradual rises may represent a new kind of state, intermediate both in timescale and in proportion of neurons participating, which gates a neuron's ability to respond to subsequent inputs.
Close appositions between the membrane of the endoplasmic reticulum (ER) and other intracellular membranes have important functions in cell physiology. These include lipid homeostasis, regulation of Ca(2+) dynamics, and control of organelle biogenesis and dynamics. Although these membrane contacts have previously been observed in neurons, their distribution and abundance have not been systematically analyzed. Here, we have used focused ion beam-scanning electron microscopy to generate 3D reconstructions of intracellular organelles and their membrane appositions involving the ER (distance ≤30 nm) in different neuronal compartments. ER-plasma membrane (PM) contacts were particularly abundant in cell bodies, with large, flat ER cisternae apposed to the PM, sometimes with a notably narrow lumen (thin ER). Smaller ER-PM contacts occurred throughout dendrites, axons, and in axon terminals. ER contacts with mitochondria were abundant in all compartments, with the ER often forming a network that embraced mitochondria. Small focal contacts were also observed with tubulovesicular structures, likely to be endosomes, and with sparse multivesicular bodies and lysosomes found in our reconstructions. Our study provides an anatomical reference for interpreting information about interorganelle communication in neurons emerging from functional and biochemical studies.
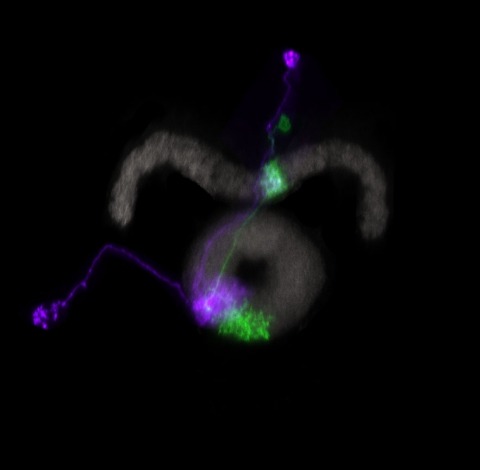
Life scientists often desire to display the signal from two different molecular probes as a single colour image, so as to convey information about the probes' relative concentrations as well as their spatial corelationship. Traditionally, such colour images are created through a merge display, where each greyscale signal is assigned to different channels of an RGB colour image. However, human perception of colour and greyscale intensity is not equivalent. Thus, a merged image display conveys to the typical viewer only a subset of the absolute and relative intensity information present in and between two greyscale images. The Commission Internationale de l'Eclairage L*a*b* colour space (CIELAB) has been designed to specify colours according to the perceptually defined quantities of hue (perceived colour) and luminosity (perceived brightness). Here, we use the CIELAB colour space to encode two dimensions of information about two greyscale images within these two perceptual dimensions of a single colour image. We term our method a Perceptually Uniform Projection display and show using biological image examples how these displays convey more information about two greyscale signals than comparable RGB colour space-based techniques.
Hippocampal place cells represent different environments with distinct neural activity patterns. Following an abrupt switch between two familiar configurations of visual cues defining two environments, the hippocampal neural activity pattern switches almost immediately to the corresponding representation. Surprisingly, during a transient period following the switch to the new environment, occasional fast transitions of activity patterns between the representations (flickering) were observed (Jezek et al. 2011). Here we show that an attractor neural network model of place cells with connections endowed with short-term synaptic plasticity can account for this phenomenon. A memory trace of the recent history of network activity is maintained in the state of the synapses, allowing the network to temporarily reactivate the representation of the previous environment in the absence of the corresponding sensory cues. The model predicts that the number of flickering events depends on the amplitude of the ongoing theta rhythm and the distance between the current position of the animal and its position at the time of cue switching. We test these predictions with new analysis of experimental data. These results suggest a potential role of short-term synaptic plasticity in recruiting the activity of different cell assemblies and in shaping hippocampal activity of behaving animals. This article is protected by copyright. All rights reserved.
The organization of the eukaryotic cell into discrete membrane-bound organelles allows for the separation of incompatible biochemical processes, but the activities of these organelles must be coordinated. For example, lipid metabolism is distributed between the endoplasmic reticulum for lipid synthesis, lipid droplets for storage and transport, mitochondria and peroxisomes for β-oxidation, and lysosomes for lipid hydrolysis and recycling. It is increasingly recognized that organelle contacts have a vital role in diverse cellular functions. However, the spatial and temporal organization of organelles within the cell remains poorly characterized, as fluorescence imaging approaches are limited in the number of different labels that can be distinguished in a single image. Here we present a systems-level analysis of the organelle interactome using a multispectral image acquisition method that overcomes the challenge of spectral overlap in the fluorescent protein palette. We used confocal and lattice light sheet instrumentation and an imaging informatics pipeline of five steps to achieve mapping of organelle numbers, volumes, speeds, positions and dynamic inter-organelle contacts in live cells from a monkey fibroblast cell line. We describe the frequency and locality of two-, three-, four- and five-way interactions among six different membrane-bound organelles (endoplasmic reticulum, Golgi, lysosome, peroxisome, mitochondria and lipid droplet) and show how these relationships change over time. We demonstrate that each organelle has a characteristic distribution and dispersion pattern in three-dimensional space and that there is a reproducible pattern of contacts among the six organelles, that is affected by microtubule and cell nutrient status. These live-cell confocal and lattice light sheet spectral imaging approaches are applicable to any cell system expressing multiple fluorescent probes, whether in normal conditions or when cells are exposed to disturbances such as drugs, pathogens or stress. This methodology thus offers a powerful descriptive tool and can be used to develop hypotheses about cellular organization and dynamics.
Many animals maintain an internal representation of their heading as they move through their surroundings. Such a compass representation was recently discovered in a neural population in the Drosophila melanogaster central complex, a brain region implicated in spatial navigation. Here, we use two-photon calcium imaging and electrophysiology in head-fixed walking flies to identify a different neural population that conjunctively encodes heading and angular velocity, and is excited selectively by turns in either the clockwise or counterclockwise direction. We show how these mirror-symmetric turn responses combine with the neurons' connectivity to the compass neurons to create an elegant mechanism for updating the fly's heading representation when the animal turns in darkness. This mechanism, which employs recurrent loops with an angular shift, bears a resemblance to those proposed in theoretical models for rodent head direction cells. Our results provide a striking example of structure matching function for a broadly relevant computation.
Advances in fluorescence microscopy promise to unlock details of biological systems with high spatiotemporal precision. These new techniques also place a heavy demand on the 'photon budget'-the number of photons one can extract from a sample. Improving the photostability of small molecule fluorophores using chemistry is a straightforward method for increasing the photon budget. Here, we review the (sometimes sparse) efforts to understand the mechanism of fluorophore photobleaching and recent advances to improve photostability through reducing the propensity for oxidation or through intramolecular triplet-state quenching. Our intent is to inspire a more thorough mechanistic investigation of photobleaching and the use of precise chemistry to improve fluorescent probes.
New work on innate escape behavior shows that mice spontaneously form a spatially precise memory of the location of shelter, which is laid down quickly and updated continuously.
The insect juvenile hormone receptor is a basic helix-loop-helix (bHLH), Per-Arnt-Sim (PAS) domain protein, a novel type of hormone receptor. In higher flies like Drosophila, the ancestral receptor germ cell-expressed (gce) gene has duplicated to yield the paralog Methoprene-tolerant (Met). These paralogous receptors share redundant function during development but play unique roles in adults. Some aspects of JH function apparently require one receptor or the other. To provide a foundation for studying JH receptor function, we have recapitulated endogenous JH receptor expression with single cell resolution. Using Bacteria Artificial Chromosome (BAC) recombineering and a transgenic knock-in, we have generated a spatiotemporal expressional atlas of Metand gce throughout development. We demonstrate JH receptor expression in known JH target tissues, in which temporal expression corresponds with periods of hormone sensitivity. Larval expression largely supports the notion of functional redundancy. Furthermore, we provide the neuroanatomical distribution of JH receptors in both the larval and adult central nervous system, which will serve as a platform for future studies regarding JH action on insect behavior.
High-resolution serial-section electron microscopy (ssEM) makes it possible to investigate the dense meshwork of axons, dendrites, and synapses that form neuronal circuits. However, the imaging scale required to comprehensively reconstruct these structures is more than ten orders of magnitude smaller than the spatial extents occupied by networks of interconnected neurons, some of which span nearly the entire brain. Difficulties in generating and handling data for large volumes at nanoscale resolution have thus restricted vertebrate studies to fragments of circuits. These efforts were recently transformed by advances in computing, sample handling, and imaging techniques, but high-resolution examination of entire brains remains a challenge. Here, we present ssEM data for the complete brain of a larval zebrafish (Danio rerio) at 5.5 days post-fertilization. Our approach utilizes multiple rounds of targeted imaging at different scales to reduce acquisition time and data management requirements. The resulting dataset can be analysed to reconstruct neuronal processes, permitting us to survey all myelinated axons (the projectome). These reconstructions enable precise investigations of neuronal morphology, which reveal remarkable bilateral symmetry in myelinated reticulospinal and lateral line afferent axons. We further set the stage for whole-brain structure-function comparisons by co-registering functional reference atlases and in vivo two-photon fluorescence microscopy data from the same specimen. All obtained images and reconstructions are provided as an open-access resource.