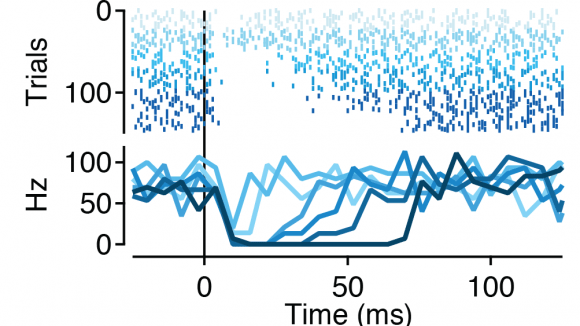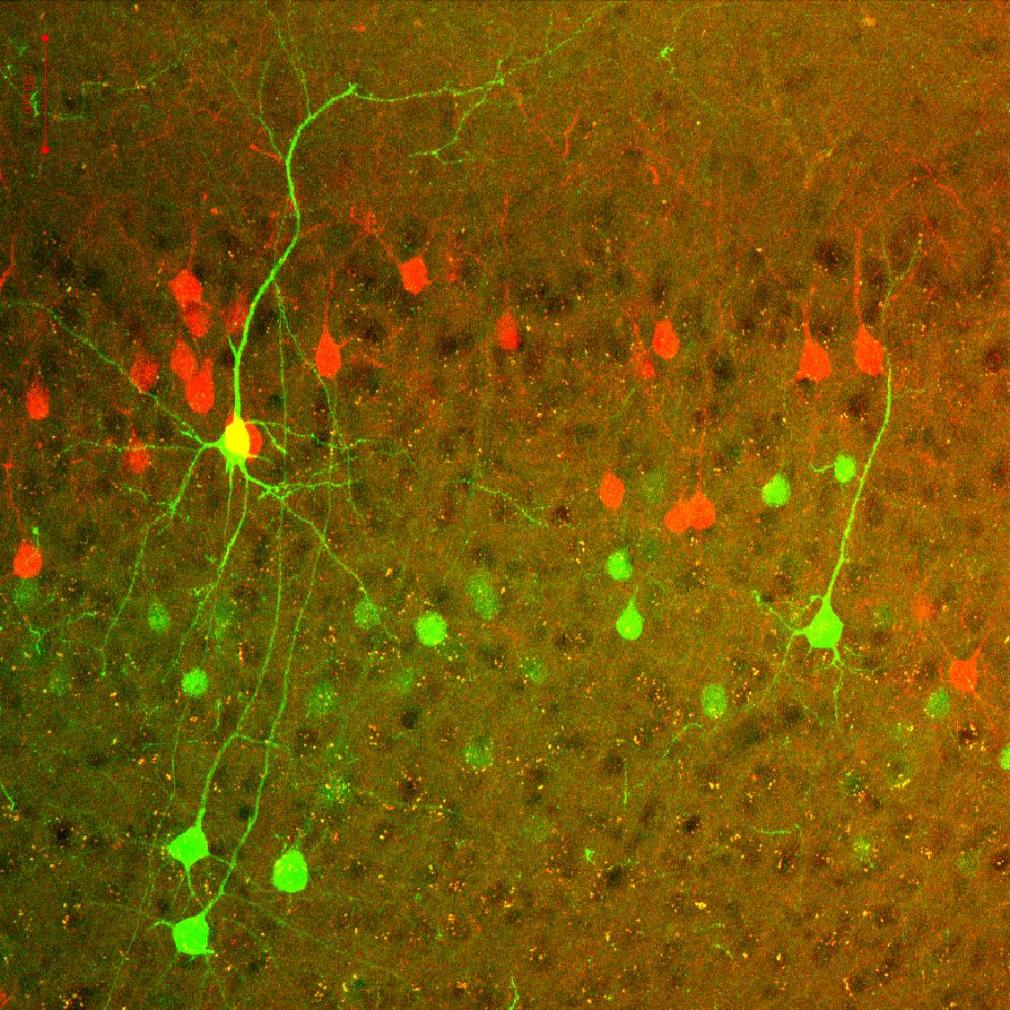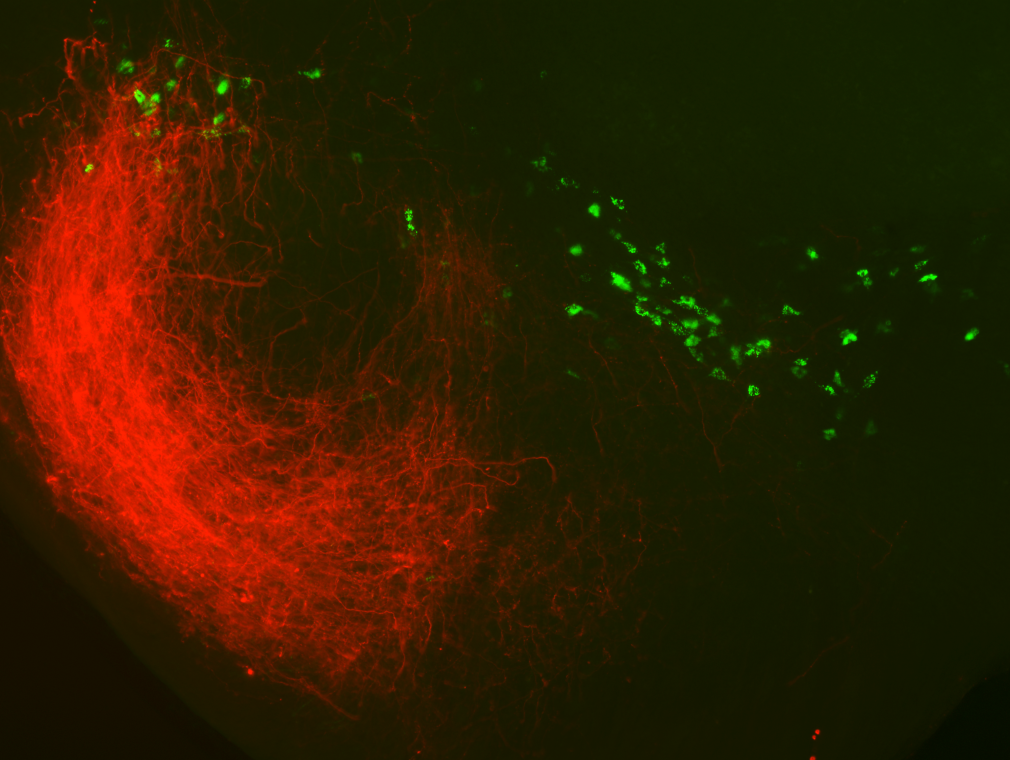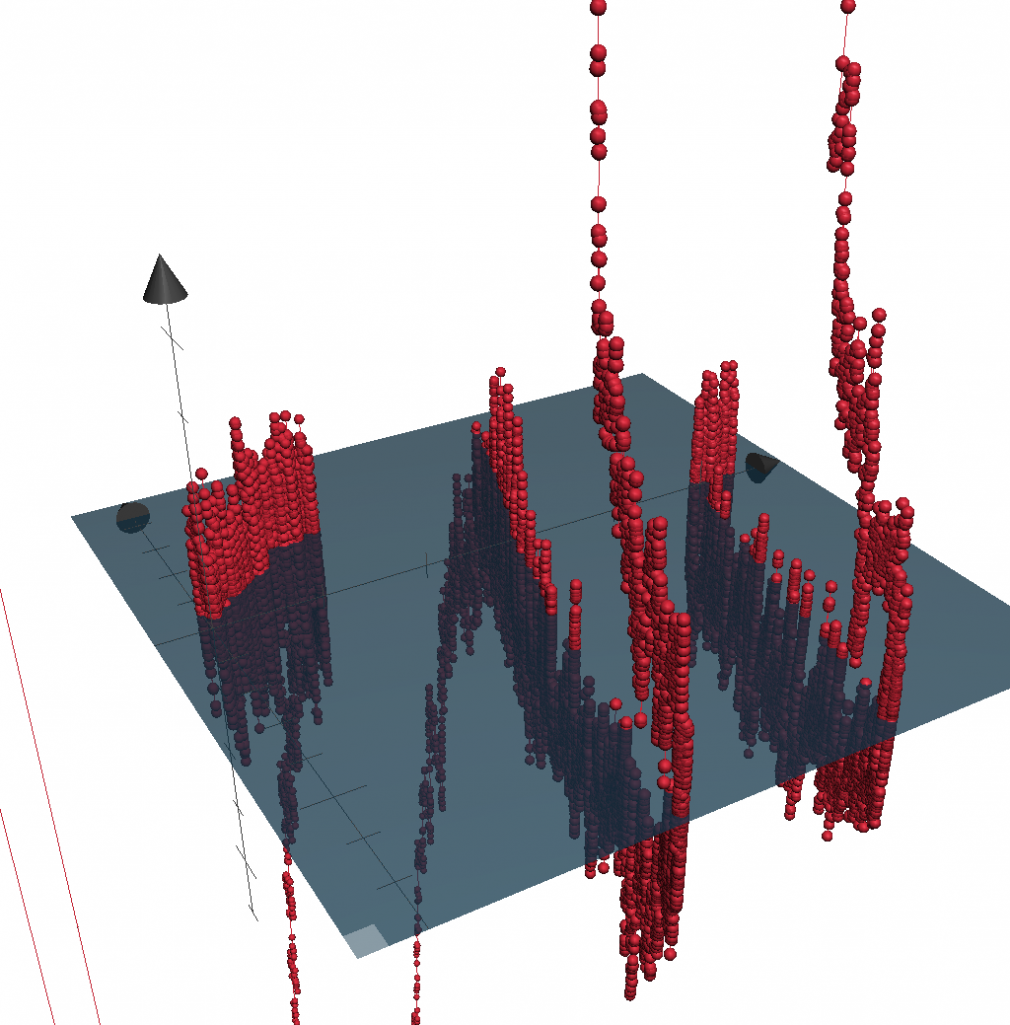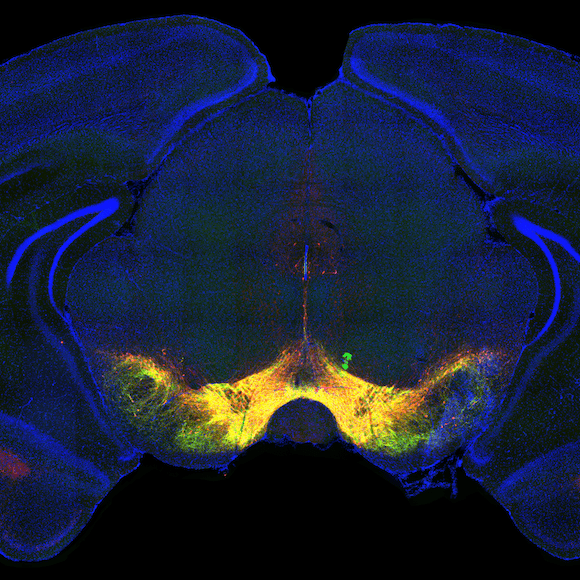
Voluntary, purposive behavior requires that we extract information about the world, formulate plans for action, and then execute the movements required to bring about desired outcomes. Our lab studies a critical nexus in the mammalian brain where sensory information and motor planning come together to subserve volition - the basal ganglia.
Lab Updates
Our recent paper described a behavioral task in which mice learn to maximize reward by adopting optimal waiting times in the presence of highly variable reward delays.
Our new paper describing how the inhibitory microcircuit in the midbrain determines the gain of the basal ganglia output is now in press.
We have developed a system for rapidly configurable and switchable mechanical system for in vivo and in vitro electrophysiology and imaging.
The work in our lab aims to elucidate the neurobiology of purposive behavior, as well as its pathological disruption in Parkinson's disease and addiction. See all the updates from the lab.
Our lab spans a wide range of technical approaches from electrophysiology during behavior to two-photon imaging. For our work we seek talented people with skills in physiology, behavior, imaging, or, computation, and a strong desire to combine multiple technical approaches. Inquiries can be sent directly to the lab by contacting us.
For interested students, we have a small number of slots for exceptional graduate students through the Janelia Graduate Program or the Graduate Research Fellowship program. For undergraduates, our lab is a regular participant in the Janelia Undergraduate Scholars program.
Published work
Over the past few years we have focused on studying the circuits of the basal ganglia in the context of reward seeking behaviors in mice. Previous work has spanned a range of techniques and questions from behavioral measures of disease progression in human patients to the atomic structure of glutamate receptors.
Data derived from: Google Scholar.
Title
~100 year-1
"...The problem of actions, in other words the effect which the organism is striving to achieve, is something which is not yet, but which is due to be brought about. The problem of action, thus, is the reflection or model of future requirements (somehow coded in the brain); and a vitally useful or significant action cannot be either programmed or accomplished if the brain has not created a prerequisite directive in the form of future requirements that we have just mentioned."
-N. Bernstein (1962)