Main Menu (Mobile)- Block
- Our Research
-
Support Teams
- Overview
- Anatomy and Histology
- Cryo-Electron Microscopy
- Electron Microscopy
- Flow Cytometry
- Fly Facility
- Gene Targeting and Transgenics
- Immortalized Cell Line Culture
- Integrative Imaging
- Janelia Experimental Technology
- Mass Spectrometry
- Media Prep
- Molecular Genomics
- Primary & iPS Cell Culture
- Project Pipeline Support
- Project Technical Resources
- Quantitative Genomics
- Scientific Computing Software
- Scientific Computing Systems
- Viral Tools
- Vivarium
- Open Science
- You + Janelia
- About Us
Main Menu - Block
- Overview
- Anatomy and Histology
- Cryo-Electron Microscopy
- Electron Microscopy
- Flow Cytometry
- Fly Facility
- Gene Targeting and Transgenics
- Immortalized Cell Line Culture
- Integrative Imaging
- Janelia Experimental Technology
- Mass Spectrometry
- Media Prep
- Molecular Genomics
- Primary & iPS Cell Culture
- Project Pipeline Support
- Project Technical Resources
- Quantitative Genomics
- Scientific Computing Software
- Scientific Computing Systems
- Viral Tools
- Vivarium

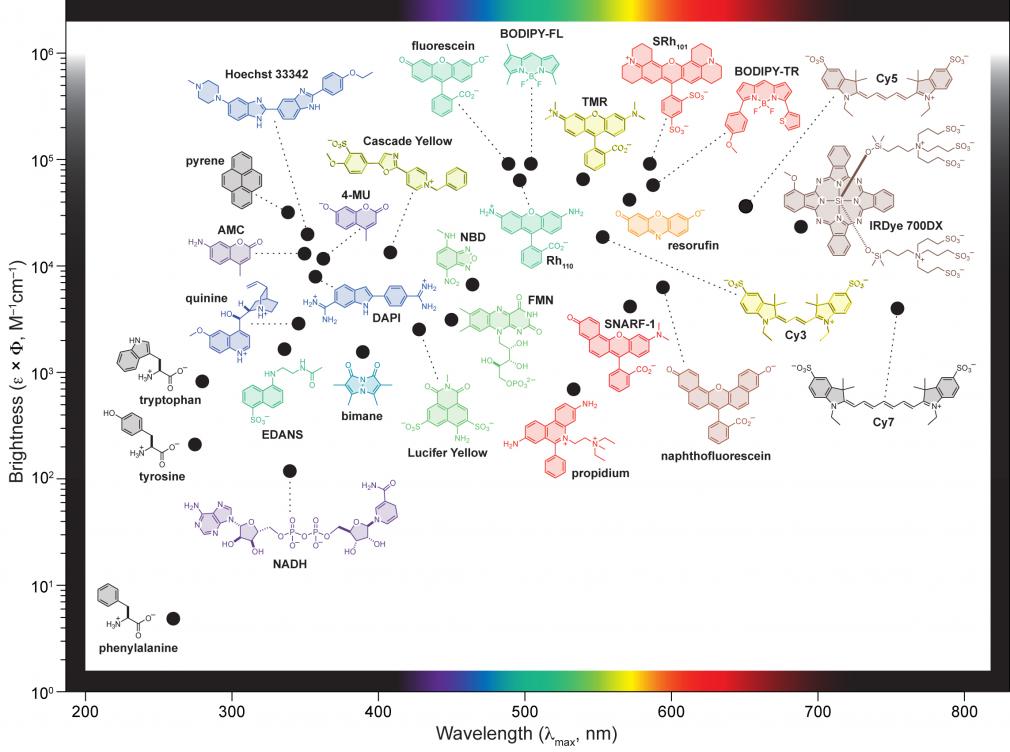
We work at the interface of chemistry and biology, assembling small molecule fluorescent dyes that facilitate sophisticated biological studies.
We are interested in designing and building small molecules to measure or manipulate biological systems. Our laboratory synthesizes bright fluorescent labels that enable the imaging of individual molecules in living cells. We also develop fluorogenic probes where the chemical and photophysical properties can be masked by assorted molecular functionalities and then unmasked by a user-designated process involving light, enzymatic activity, or environmental changes. This chemical masking suppresses unwanted fluorescence signals, thereby functioning as a filter for bioimaging and other experiments. Combining these novel compounds with advances in instrumentation, protein engineering, and genetic manipulation allows us to devise sophisticated ways to illuminate complex biological systems.
We use modern organic chemistry to transform old dyes into tools for 21st century biology.
-Luke Lavis
A review of small molecule fluorescent dyes
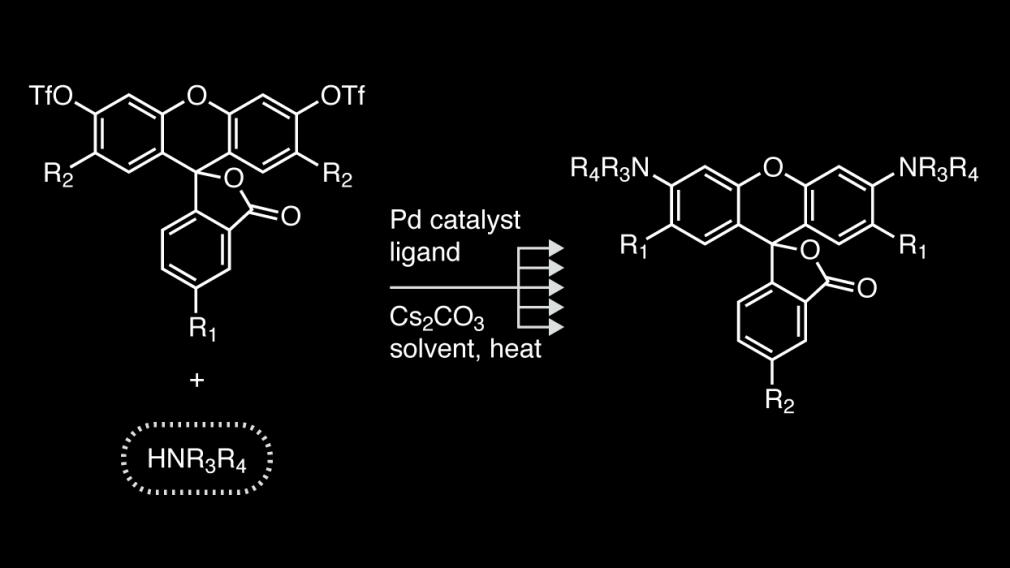
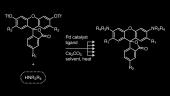
Read the PaperOur lab has established a simple method to synthesize a diverse array of rhodamine dyes from simple fluorescein compounds.
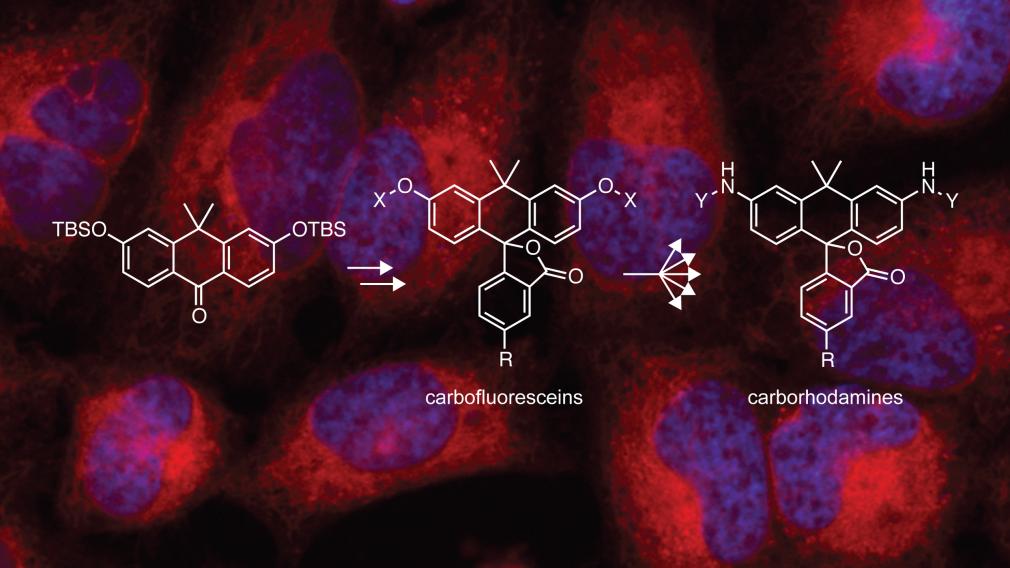
Read the paperWe have developed an efficient synthesis of the carbon-containing analogs of fluorescein and rhodamine. These compounds constitute a new high-contrast scaffold for fluorogenic molecules.
Read the paperWe have demonstrated that a simple selective esterase–ester pair can allow the targeted delivery of dyes and drugs to genetically defined subsets of cells.
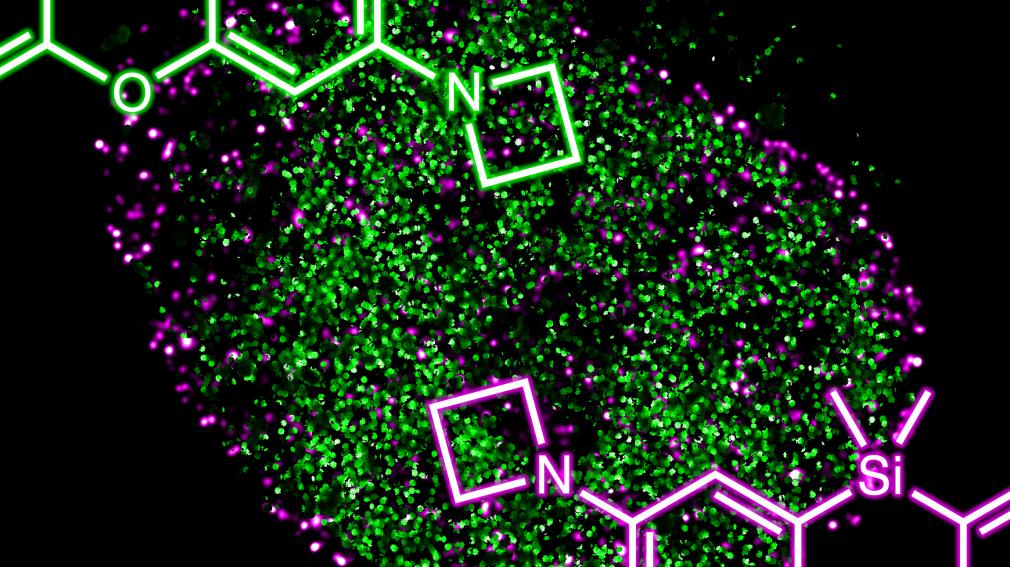
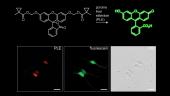
Read the paperReplacing ubiquitous dimethylamino groups found in a variety of fluorophores with the azetidine functionality gives large increases in brightness with a small change in chemical structure. This allows sophisticated cellular imaging experiments such as multicolor single-molecule microscopy.
Read the paper