Main Menu (Mobile)- Block
- Our Research
-
Support Teams
- Overview
- Anatomy and Histology
- Cryo-Electron Microscopy
- Electron Microscopy
- Flow Cytometry
- Fly Facility
- Gene Targeting and Transgenics
- Immortalized Cell Line Culture
- Integrative Imaging
- Janelia Experimental Technology
- Mass Spectrometry
- Media Prep
- Molecular Genomics
- Primary & iPS Cell Culture
- Project Pipeline Support
- Project Technical Resources
- Quantitative Genomics
- Scientific Computing Software
- Scientific Computing Systems
- Viral Tools
- Vivarium
- Open Science
- You + Janelia
- About Us
Main Menu - Block
- Overview
- Anatomy and Histology
- Cryo-Electron Microscopy
- Electron Microscopy
- Flow Cytometry
- Fly Facility
- Gene Targeting and Transgenics
- Immortalized Cell Line Culture
- Integrative Imaging
- Janelia Experimental Technology
- Mass Spectrometry
- Media Prep
- Molecular Genomics
- Primary & iPS Cell Culture
- Project Pipeline Support
- Project Technical Resources
- Quantitative Genomics
- Scientific Computing Software
- Scientific Computing Systems
- Viral Tools
- Vivarium
We aim to connect our understanding of neurons, synapses, and circuits to a deeper understanding of learned intelligent behavior. We obtain high resolution, large scale data from behaving animals to constrain quantitative models and ultimately theories that explain brain function.
Lab Updates
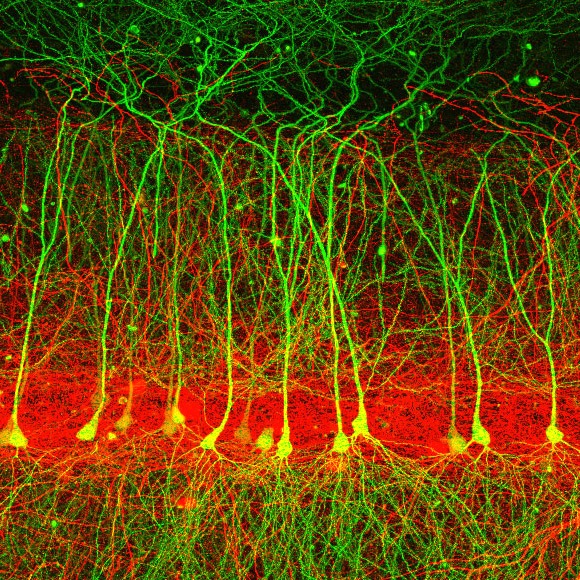
We study the function of neurons and glia in the hippocampus. This structure is a nexus of behaviorally relevant information arriving from many brain areas. These external representations are combined with internal representations of recent experiences—short-term memories—to generate output that is distributed to other brain areas, in order to adaptively guide behavior and form long-term memories. The goal of our lab is to understand how different hippocampal cell types communicate with their synaptic partners within and outside of the hippocampal circuit, altering their activity and connections as a function of experience.
The brain seems to be made up of a bewildering complexity of parts, and the cells within the parts seem to be characterized by an inscrutable complexity of form, extent, and relationships with each other.
Gordon M. Shepherd
The Synaptic Organization of the Brain, 2nd ed., 1979.