Seventeen years ago, at a restaurant in Boulder, Colorado, three scientists leaned over a table, deep in discussion about a sketch they’d just scribbled onto the back of a napkin. It didn’t show a biochemical pathway or molecular motif, but an idea for a facility where researchers could develop and access advanced technologies.
After three years of intense discussion and planning, this modest idea developed into a philosophical blueprint of an experiment that had the potential to shift the basic tenets of scientific research. The idea was a big one – a multimillion-dollar undertaking that would turn the traditionally siloed, grant-dependent approach to research on its head.
Working with distinguished architects, HHMI’s leadership began to chart physical, and cultural, plans for a building that could house their evolving “experiment.” By October of 2006, those plans became a reality, and the doors officially opened at HHMI’s first-ever independent research institute, Janelia Research Campus. The 700-acre institute focuses on imaging technology and neurobiology, tackling what HHMI Vice President and Janelia Executive Director Gerry Rubin calls one of the last frontiers of biology: the evolutionarily conserved mechanisms that underlie how the brain works.
Now, nearly 600 scientists, 1,050 publications, and 5 million cups of coffee later, the 10th anniversary of Janelia’s opening has arrived.
“We started as, and continue to be, an experiment in the social engineering of research communities,” says Rubin.
Janelia’s launch garnered a lot of attention. Onlookers from academia met the new campus with raised eyebrows – some out of excitement, others out of skepticism. And underlying the attention and commitment was one clear question: Would it work?
“Our development is far from finished, but after 10 years, we’ve gained momentum and have found our footing in the scientific landscape,” says Rubin.
The past decade witnessed many scientific achievements and ushered in new mechanisms for fostering collaboration on an international level, such as the Visiting Scientist Program, and for facilitating open access to new technologies developed and built in-house, such as the Advanced Imaging Center. Now, as the campus continues to grow and evolve, new questions are emerging.
“We need to ask, how do we continue to challenge ourselves to do science that wouldn’t be done elsewhere? How do we get out of our comfort zone? Those questions are important in keeping momentum for any organization,” says Rubin. “It’s something we actively think about, and we want to reflect that in our choice of scientific problems and in how we approach them.”
Mapping the Connections
Ultimately, Janelia’s continuing evolution comes down to its community – scientists with insatiable curiosity and the gumption to pursue high-risk, high-reward research.
Michael Reiser, a neurobiologist and group leader at Janelia, falls into that category.
Reiser, who joined Janelia in its early days, studies the neural pathways that give rise to animal behavior. His research centers on how the brain of the fruit fly – Drosophila – processes visual stimuli while the animal navigates its environment. Take, for instance, a fly that encounters an obstacle during flight and veers to avoid a collision. Reiser and his lab aim to help build a neurological map, or “connectome,” that shows the neuron-to-neuron connections that encode these visual cues – a tree or a wall, for example – and inform the fly to alter its route. But while the team works to contribute new pieces to the neural map, their main goal centers on interpreting and explaining the part of the connectome that senses obstacles, encodes the information, and ultimately, tells the fly to swerve.
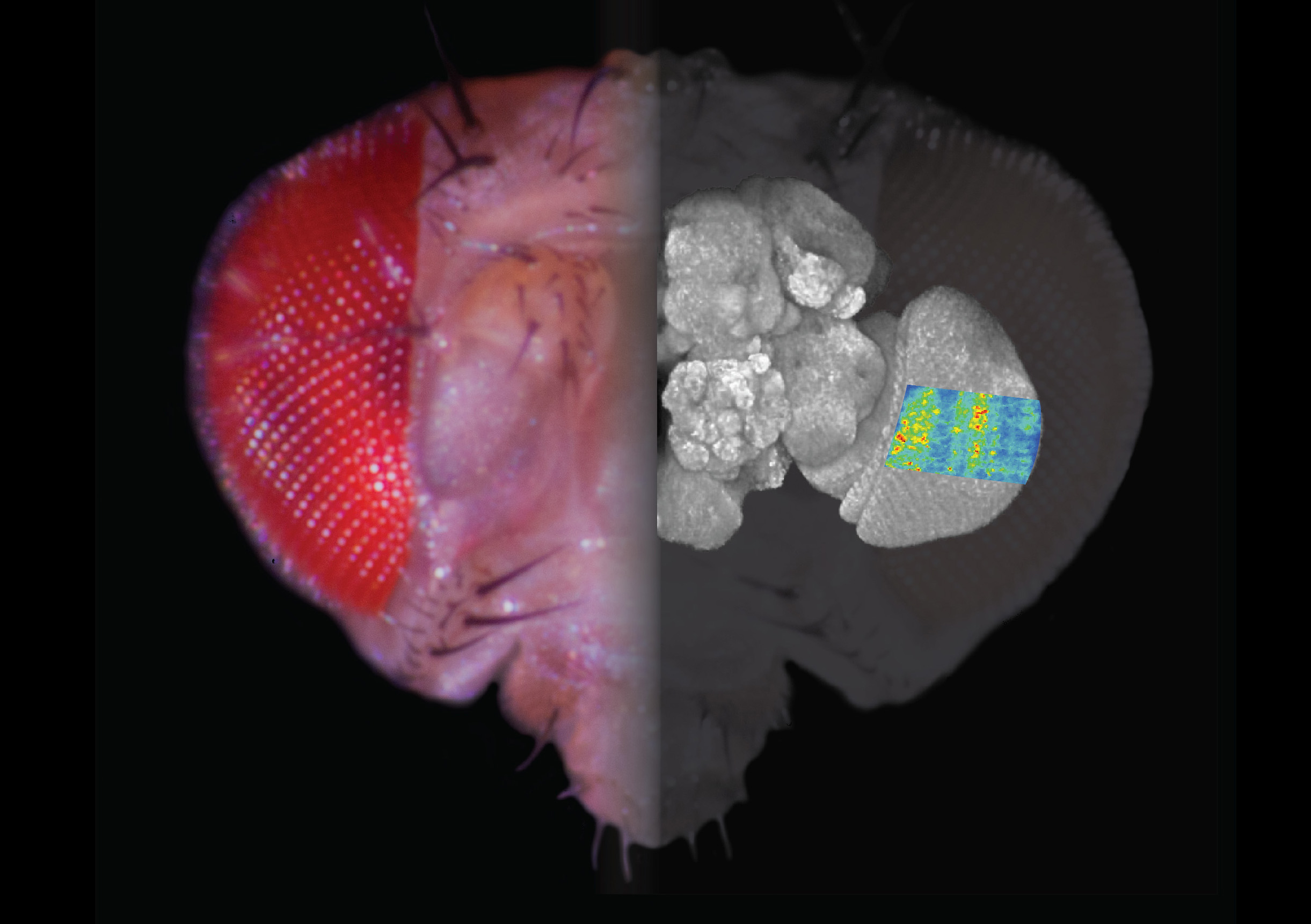
This composite image shows a fly’s head, a projection of its brain, and a two-photon calcium image of stimulus-provoked responses in the visual system.
“Trying to understand the connectome is like solving multiple puzzles at the same time,” says Reiser. “There isn’t just one experiment that really nails down any one question. We have to attack this problem from all sides.”
Fortunately, Reiser has company. He is one of many Janelians, including physicist and group leader Harald Hess, who are working to generate a Drosophila connectome. Hess, unlike Reiser, doesn’t directly interfere with fruit fly flight patterns in search of understanding the neural mechanisms. He develops tools – microscopes that allow scientists like Reiser, and many others, to witness cells in their natural context, decipher their structural details, and even inspect the dynamic processes inside a single cell.
One instrument Hess developed that captures such minutiae is the focused ion beam-scanning electron microscope, or FIB-SEM. The microscope images extremely thin layers of a fly’s brain at high resolution and generates videos, akin to flip books, from hundreds of thousands of photos that reveal neuron-neuron interactions. The images result from a two-step process: The microscope’s ion beam shears off a few nanometers of tissue to expose a new layer of the brain, in much the same way a sandblaster would strip a layer of paint from a chair. With the new layer bared, the microscope takes a picture, and the cycle continues.
The above video shows footage of the Drosophila optic lobe, capture over three months by the FIB-SEM microscope.
Hess describes Reiser as the “perfect customer” for the resulting videos – particularly those that reveal neurons in the visual system. Paired with genetic techniques that target specific neuron populations, the FIB-SEM images help Reiser and his team figure out the neural underpinnings of Drosophila behavior.
Hess believes that the fluid collaboration at Janelia is a boon to the campus’s research overall. “A major strength is not being too broadly committed,” he says. “At many other institutions you have to cover sample prep, and you need to be the surgeon, the microscopist, and the analyst. But here, there’s a comfort in knowing that I can focus on my own specialty, push that research to greater depth, and rely on other collaborators to likewise expand on their own research frontiers.”
Into the Cell
Hess and like-minded inventors at Janelia strive to push past the traditional constraints of microscopy, discovering new insights into areas like connectomics, subcellular organization, and live-cell imaging. But as with any progress in science, providing accessible information and resources to the greater scientific community is key.
Through Janelia’s Advanced Imaging Center, a selection of tools developed by Hess and fellow Janelia physicists benefit resident scientists and visitors alike.
Jennifer Lippincott-Schwartz, a group leader and cell biologist who recently moved her laboratory from the National Institutes of Health (NIH) to Janelia, has worked with Hess and his tools as both a visiting and a resident scientist. She describes their partnership as a two-way street. Hess and other close collaborators, such as group leader and Nobel Laureate Eric Betzig, ask Lippincott-Schwartz to try out new technologies and impart biologically relevant feedback. In turn, Lippincott-Schwartz approaches them with new cellular pursuits, inquiring about microscopes suited for her experiments.
In October 2016, the three researchers contributed to a paper in Science that provided new insights into one of the cell’s most prominent organelles (compartmentalized structures inside the cell, similar to internal organs in humans). Prior to the paper’s publication, scientists believed that this organelle, known as the endoplasmic reticulum, or ER, had a smooth, sheet-like appearance. Using Hess and Betzig’s custom-designed microscopes, Lippincott-Schwartz revealed that the true architecture of the ER involves tight, cross-linked tubular structures that create a flexible meshwork.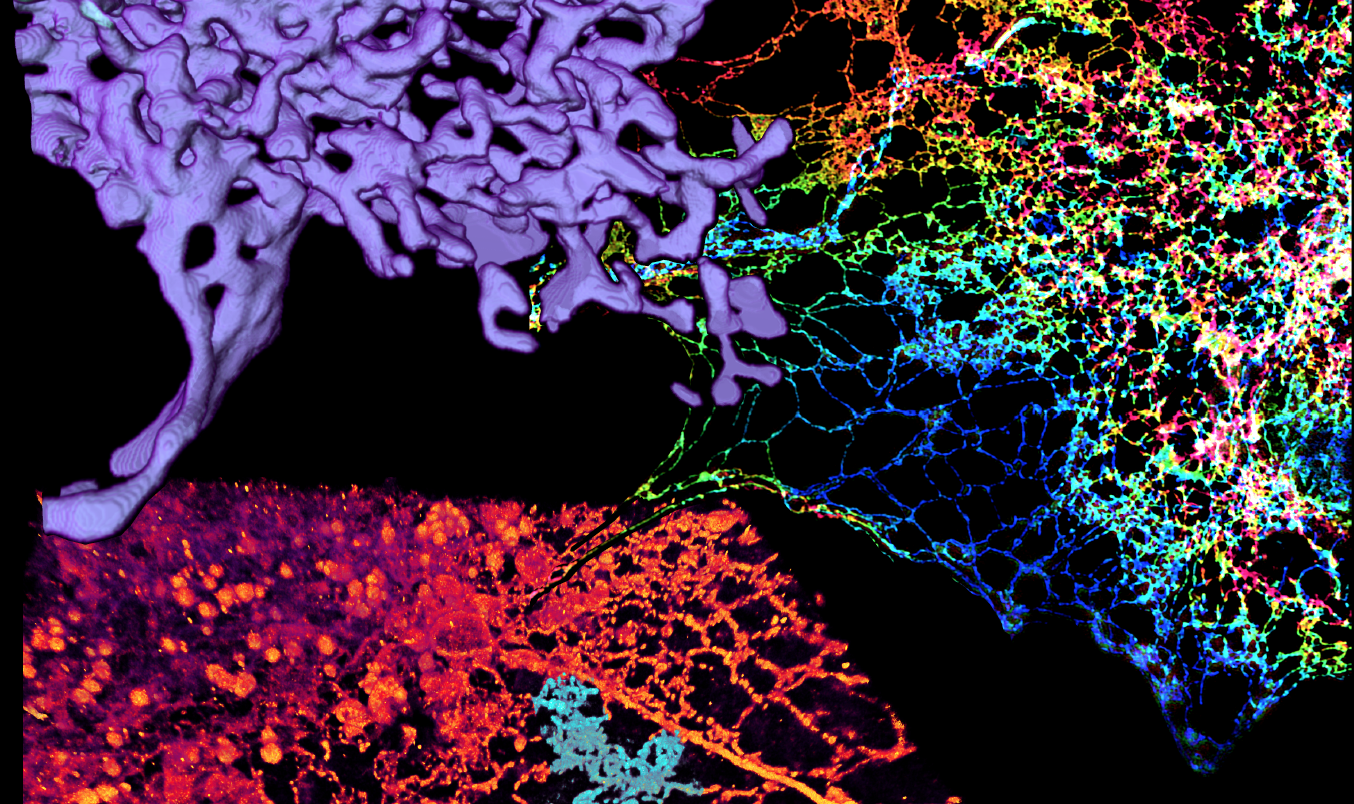
“With Eric’s microscopes we got close to getting our paper accepted, but the reviewers wanted to see electron microscopy too, which is not an easy shift if you’ve been doing a lot of light microscopy,” says Lippincott-Schwartz. “But we were able to work with Harald and the spectacular FIB-SEM and image the ER in a very short period of time. It turned out to be a tour de force of our paper.”
What’s more, the new understanding allowed Lippincott-Schwartz to build on an existing body of knowledge. Craig Blackstone and scientists at the NIH have shown that mutations in genes responsible for ER morphology are linked to spastic paraplegia in humans. The mutations cause malformation of the ER, which hampers the organelle’s ability to function properly in neurons, causing them to die. The resulting nerve damage leads to muscular dysfunction in the lower limbs. Lippincott-Schwartz’s paper suggested a link between the mutations and ER failure, and ultimately nerve cell failure, in the disease.
While the group’s paper does have medical implications, their fundamental objective as researchers and tool designers is not to hunt for treatments to neurological disorders. Likewise, for Reiser’s team – and Janelia as a whole – the goal of their research is not to cure brain diseases, but to help propel neuroscience as a field. The hope is to forge the groundwork that will empower medical advances and help physicians better understand, and therefore treat, neurological disorders in the future.
“A lot of what we do now is only possible because of the discoveries from 10, 20, or 50 years ago – discoveries made by people who didn’t realize their contributions would be relevant to real-world problems,” says Rubin. “We’re a medical institute, and I believe everything that we do will be medically relevant – maybe not for me, or for you, but perhaps for my seven-year-old granddaughter. It’s just a matter of time.”
Discover Janelia
Interested in learning more about how HHMI turned a quiet, wooded hillside along the Potomac River into a cutting-edge research facility?
The Archives
Explore Janelia's history through a curated collection of scientific and cultural reference points.


