Filter
Associated Lab
- Ahrens Lab (3) Apply Ahrens Lab filter
- Aso Lab (6) Apply Aso Lab filter
- Baker Lab (2) Apply Baker Lab filter
- Betzig Lab (11) Apply Betzig Lab filter
- Branson Lab (6) Apply Branson Lab filter
- Cardona Lab (5) Apply Cardona Lab filter
- Chklovskii Lab (2) Apply Chklovskii Lab filter
- Cui Lab (5) Apply Cui Lab filter
- Darshan Lab (1) Apply Darshan Lab filter
- Dickson Lab (2) Apply Dickson Lab filter
- Druckmann Lab (3) Apply Druckmann Lab filter
- Dudman Lab (3) Apply Dudman Lab filter
- Eddy/Rivas Lab (4) Apply Eddy/Rivas Lab filter
- Egnor Lab (1) Apply Egnor Lab filter
- Fetter Lab (5) Apply Fetter Lab filter
- Fitzgerald Lab (3) Apply Fitzgerald Lab filter
- Freeman Lab (7) Apply Freeman Lab filter
- Funke Lab (1) Apply Funke Lab filter
- Gonen Lab (5) Apply Gonen Lab filter
- Grigorieff Lab (8) Apply Grigorieff Lab filter
- Harris Lab (7) Apply Harris Lab filter
- Heberlein Lab (2) Apply Heberlein Lab filter
- Hess Lab (7) Apply Hess Lab filter
- Jayaraman Lab (4) Apply Jayaraman Lab filter
- Ji Lab (4) Apply Ji Lab filter
- Karpova Lab (1) Apply Karpova Lab filter
- Keleman Lab (2) Apply Keleman Lab filter
- Keller Lab (8) Apply Keller Lab filter
- Lavis Lab (5) Apply Lavis Lab filter
- Leonardo Lab (2) Apply Leonardo Lab filter
- Li Lab (3) Apply Li Lab filter
- Lippincott-Schwartz Lab (4) Apply Lippincott-Schwartz Lab filter
- Liu (Zhe) Lab (4) Apply Liu (Zhe) Lab filter
- Looger Lab (9) Apply Looger Lab filter
- Magee Lab (5) Apply Magee Lab filter
- Menon Lab (2) Apply Menon Lab filter
- Murphy Lab (1) Apply Murphy Lab filter
- Otopalik Lab (2) Apply Otopalik Lab filter
- Pachitariu Lab (2) Apply Pachitariu Lab filter
- Pastalkova Lab (3) Apply Pastalkova Lab filter
- Reiser Lab (2) Apply Reiser Lab filter
- Romani Lab (4) Apply Romani Lab filter
- Rubin Lab (16) Apply Rubin Lab filter
- Saalfeld Lab (4) Apply Saalfeld Lab filter
- Scheffer Lab (2) Apply Scheffer Lab filter
- Schreiter Lab (4) Apply Schreiter Lab filter
- Sgro Lab (3) Apply Sgro Lab filter
- Shroff Lab (1) Apply Shroff Lab filter
- Simpson Lab (4) Apply Simpson Lab filter
- Singer Lab (16) Apply Singer Lab filter
- Spruston Lab (7) Apply Spruston Lab filter
- Stern Lab (4) Apply Stern Lab filter
- Sternson Lab (7) Apply Sternson Lab filter
- Svoboda Lab (9) Apply Svoboda Lab filter
- Tebo Lab (2) Apply Tebo Lab filter
- Tillberg Lab (1) Apply Tillberg Lab filter
- Tjian Lab (6) Apply Tjian Lab filter
- Truman Lab (6) Apply Truman Lab filter
- Turaga Lab (2) Apply Turaga Lab filter
- Turner Lab (3) Apply Turner Lab filter
- Wang (Shaohe) Lab (1) Apply Wang (Shaohe) Lab filter
- Wu Lab (1) Apply Wu Lab filter
- Zlatic Lab (4) Apply Zlatic Lab filter
- Zuker Lab (2) Apply Zuker Lab filter
Associated Project Team
Publication Date
- December 2015 (15) Apply December 2015 filter
- November 2015 (22) Apply November 2015 filter
- October 2015 (18) Apply October 2015 filter
- September 2015 (20) Apply September 2015 filter
- August 2015 (17) Apply August 2015 filter
- July 2015 (18) Apply July 2015 filter
- June 2015 (20) Apply June 2015 filter
- May 2015 (20) Apply May 2015 filter
- April 2015 (24) Apply April 2015 filter
- March 2015 (21) Apply March 2015 filter
- February 2015 (36) Apply February 2015 filter
- January 2015 (21) Apply January 2015 filter
- Remove 2015 filter 2015
Type of Publication
252 Publications
Showing 71-80 of 252 resultsDendritic integration of synaptic inputs mediates rapid neural computation as well as longer-lasting plasticity. Several channel types can mediate dendritically initiated spikes (dSpikes), which may impact information processing and storage across multiple timescales; however, the roles of different channels in the rapid vs long-term effects of dSpikes are unknown. We show here that dSpikes mediated by Nav channels (blocked by a low concentration of TTX) are required for long-term potentiation (LTP) in the distal apical dendrites of hippocampal pyramidal neurons. Furthermore, imaging, simulations, and buffering experiments all support a model whereby fast Nav channel-mediated dSpikes (Na-dSpikes) contribute to LTP induction by promoting large, transient, localized increases in intracellular calcium concentration near the calcium-conducting pores of NMDAR and L-type Cav channels. Thus, in addition to contributing to rapid neural processing, Na-dSpikes are likely to contribute to memory formation via their role in long-lasting synaptic plasticity.
Chronic immobilization stress (CIS) shortens apical dendritic trees of CA3 pyramidal neurons in the hippocampus of the male rat, and dendritic length may be a determinant of vulnerability to stress. Expression of the polysialylated form of neural cell adhesion molecule (PSA-NCAM) in the hippocampal formation is increased by stress, while PSA removal by Endo-neuraminidase-N (endo-N) is known to cause the mossy fibers to defasciculate and synapse ectopically in their CA3 target area. We show here that enzymatic removal of PSA produced a remarkable expansion of dendritic arbors of CA3 pyramidal neurons, with a lesser effect in CA1. This expansion eclipsed the CIS-induced shortening of CA3 dendrites, with the expanded dendrites of both no-stress-endo-N and CIS-endo-N rats being longer than those in no-stress-control rats and much longer than those in CIS-control rats. As predicted by the hypothesis that endo-N-induced dendritic expansion might increase vulnerability to excitotoxic challenge, systemic injection with kainic acid, showed markedly increased neuronal degeneration, as assessed by fluorojade B histochemistry, in rats that had been treated with endo-N compared to vehicle-treated rats throughout the entire hippocampal formation. PSA removal also exacerbated the CIS-induced reduction in body weight and abolished effects of CIS on NPY and NR2B mRNA levels. These findings support the hypothesis that CA3 arbor plasticity plays a protective role during prolonged stress and clarify the role of PSA-NCAM in stress-induced dendritic plasticity.
We describe a general approach to designing two-dimensional (2D) protein arrays mediated by noncovalent protein-protein interfaces. Protein homo-oligomers are placed into one of the seventeen 2D layer groups, the degrees of freedom of the lattice are sampled to identify configurations with shape-complementary interacting surfaces, and the interaction energy is minimized using sequence design calculations. We used the method to design proteins that self-assemble into layer groups P 3 2 1, P 4 21 2, and P 6. Projection maps of micrometer-scale arrays, assembled both in vitro and in vivo, are consistent with the design models and display the target layer group symmetry. Such programmable 2D protein lattices should enable new approaches to structure determination, sensing, and nanomaterial engineering.
Calcium signaling has long been associated with key events of immunity, including chemotaxis, phagocytosis, and activation. However, imaging and manipulation of calcium flux in motile immune cells in live animals remain challenging. Using light-sheet microscopy for in vivo calcium imaging in zebrafish, we observe characteristic patterns of calcium flux triggered by distinct events, including phagocytosis of pathogenic bacteria and migration of neutrophils toward inflammatory stimuli. In contrast to findings from ex vivo studies, we observe enriched calcium influx at the leading edge of migrating neutrophils. To directly manipulate calcium dynamics in vivo, we have developed transgenic lines with cell-specific expression of the mammalian TRPV1 channel, enabling ligand-gated, reversible, and spatiotemporal control of calcium influx. We find that controlled calcium influx can function to help define the neutrophil's leading edge. Cell-specific TRPV1 expression may have broad utility for precise control of calcium dynamics in other immune cell types and organisms.
Adaptive optics by direct imaging of the wavefront distortions of a laser-induced guide star has long been used in astronomy, and more recently in microscopy to compensate for aberrations in transparent specimens. Here we extend this approach to tissues that strongly scatter visible light by exploiting the reduced scattering of near-infrared guide stars. The method enables in vivo two-photon morphological and functional imaging down to 700 μm inside the mouse brain.
Circadian (daily) rhythms are present in almost all plants and animals. In mammals, a brain clock located in the hypothalamic suprachiasmatic nucleus maintains synchrony between environmental light/dark cycles and physiology and behavior. Over the past 100 y, especially with the advent of electric lighting, modern society has resulted in a round-the-clock lifestyle, in which natural connections between rest/activity cycles and environmental light/dark cycles have been degraded or even broken. Instances in which rapid changes to sleep patterns are necessary, such as transmeridian air travel, demonstrate negative effects of acute circadian disruption on physiology and behavior. However, the ramifications of chronic disruption of the circadian clock for mental and physical health are not yet fully understood. By housing mice in 20-h light/dark cycles, incongruous with their endogenous ∼24-h circadian period, we were able to model the effects of chronic circadian disruption noninvasively. Housing in these conditions results in accelerated weight gain and obesity, as well as changes in metabolic hormones. In the brain, circadian-disrupted mice exhibit a loss of dendritic length and decreased complexity of neurons in the prelimbic prefrontal cortex, a brain region important in executive function and emotional control. Disrupted animals show decreases in cognitive flexibility and changes in emotionality consistent with the changes seen in neural architecture. How our findings translate to humans living and working in chronic circadian disruption is unknown, but we believe that this model can provide a foundation to understand how environmental disruption of circadian rhythms impacts the brain, behavior, and physiology.
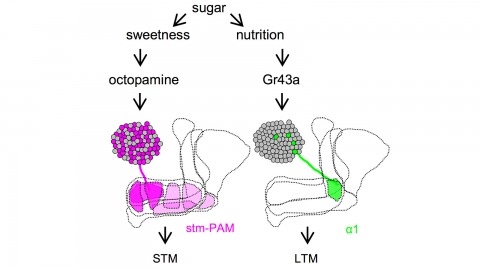
Drosophila melanogaster can acquire a stable appetitive olfactory memory when the presentation of a sugar reward and an odor are paired. However, the neuronal mechanisms by which a single training induces long-term memory are poorly understood. Here we show that two distinct subsets of dopamine neurons in the fly brain signal reward for short-term (STM) and long-term memories (LTM). One subset induces memory that decays within several hours, whereas the other induces memory that gradually develops after training. They convey reward signals to spatially segregated synaptic domains of the mushroom body (MB), a potential site for convergence. Furthermore, we identified a single type of dopamine neuron that conveys the reward signal to restricted subdomains of the mushroom body lobes and induces long-term memory. Constant appetitive memory retention after a single training session thus comprises two memory components triggered by distinct dopamine neurons.
Ionotropic glutamate receptors principally mediate fast excitatory transmission in the brain. Among the three classes of ionotropic glutamate receptors, kainate receptors (KARs) have a unique brain distribution, which has been historically defined by (3)H-radiolabeled kainate binding. Compared with recombinant KARs expressed in heterologous cells, synaptic KARs exhibit characteristically slow rise-time and decay kinetics. However, the mechanisms responsible for these distinct KAR properties remain unclear. We found that both the high-affinity binding pattern in the mouse brain and the channel properties of native KARs are determined by the KAR auxiliary subunit Neto1. Through modulation of agonist binding affinity and off-kinetics of KARs, but not trafficking of KARs, Neto1 determined both the KAR high-affinity binding pattern and the distinctively slow kinetics of postsynaptic KARs. By regulating KAR excitatory postsynaptic current kinetics, Neto1 can control synaptic temporal summation, spike generation and fidelity.
The apical tuft is the most remote area of the dendritic tree of neocortical pyramidal neurons. Despite its distal location, the apical dendritic tuft of layer 5 pyramidal neurons receives substantial excitatory synaptic drive and actively processes corticocortical input during behavior. The properties of the voltage-activated ion channels that regulate synaptic integration in tuft dendrites have, however, not been thoroughly investigated. Here, we use electrophysiological and optical approaches to examine the subcellular distribution and function of hyperpolarization-activated cyclic nucleotide-gated nonselective cation (HCN) channels in rat layer 5B pyramidal neurons. Outside-out patch recordings demonstrated that the amplitude and properties of ensemble HCN channel activity were uniform in patches excised from distal apical dendritic trunk and tuft sites. Simultaneous apical dendritic tuft and trunk whole-cell current-clamp recordings revealed that the pharmacological blockade of HCN channels decreased voltage compartmentalization and enhanced the generation and spread of apical dendritic tuft and trunk regenerative activity. Furthermore, multisite two-photon glutamate uncaging demonstrated that HCN channels control the amplitude and duration of synaptically evoked regenerative activity in the distal apical dendritic tuft. In contrast, at proximal apical dendritic trunk and somatic recording sites, the blockade of HCN channels decreased excitability. Dynamic-clamp experiments revealed that these compartment-specific actions of HCN channels were heavily influenced by the local and distributed impact of the high density of HCN channels in the distal apical dendritic arbor. The properties and subcellular distribution pattern of HCN channels are therefore tuned to regulate the interaction between integration compartments in layer 5B pyramidal neurons.
Progressive depletion of midbrain dopamine neurons (PDD) is associated with deficits in the initiation, speed, and fluidity of voluntary movement. Models of basal ganglia function focus on initiation deficits; however, it is unclear how they account for deficits in the speed or amplitude of movement (vigor). Using an effort-based operant conditioning task for head-fixed mice, we discovered distinct functional classes of neurons in the dorsal striatum that represent movement vigor. Mice with PDD exhibited a progressive reduction in vigor, along with a selective impairment of its neural representation in striatum. Restoration of dopaminergic tone with a synthetic precursor ameliorated deficits in movement vigor and its neural representation, while suppression of striatal activity during movement was sufficient to reduce vigor. Thus, dopaminergic input to the dorsal striatum is indispensable for the emergence of striatal activity that mediates adaptive changes in movement vigor. These results suggest refined intervention strategies for Parkinson’s disease.