Filter
Associated Lab
6 Janelia Publications
Showing 1-6 of 6 resultsLearning is primarily mediated by activity-dependent modifications of synaptic strength within neuronal circuits. We discovered that place fields in hippocampal area CA1 are produced by a synaptic potentiation notably different from Hebbian plasticity. Place fields could be produced in vivo in a single trial by potentiation of input that arrived seconds before and after complex spiking. The potentiated synaptic input was not initially coincident with action potentials or depolarization. This rule, named behavioral time scale synaptic plasticity, abruptly modifies inputs that were neither causal nor close in time to postsynaptic activation. In slices, five pairings of subthreshold presynaptic activity and calcium (Ca(2+)) plateau potentials produced a large potentiation with an asymmetric seconds-long time course. This plasticity efficiently stores entire behavioral sequences within synaptic weights to produce predictive place cell activity.
The dilemma that neurotheorists face is that (1) detailed biophysical models that can be constrained by direct measurements, while being of great importance, offer no immediate insights into cognitive processes in the brain, and (2) high-level abstract cognitive models, on the other hand, while relevant for understanding behavior, are largely detached from neuronal processes and typically have many free, experimentally unconstrained parameters that have to be tuned to a particular data set and, hence, cannot be readily generalized to other experimental paradigms. In this contribution, we propose a set of "first principles" for neurally inspired cognitive modeling of memory retrieval that has no biologically unconstrained parameters and can be analyzed mathematically both at neuronal and cognitive levels. We apply this framework to the classical cognitive paradigm of free recall. We show that the resulting model accounts well for puzzling behavioral data on human participants and makes predictions that could potentially be tested with neurophysiological recording techniques.
Hippocampal place cells represent different environments with distinct neural activity patterns. Following an abrupt switch between two familiar configurations of visual cues defining two environments, the hippocampal neural activity pattern switches almost immediately to the corresponding representation. Surprisingly, during a transient period following the switch to the new environment, occasional fast transitions of activity patterns between the representations (flickering) were observed (Jezek et al. 2011). Here we show that an attractor neural network model of place cells with connections endowed with short-term synaptic plasticity can account for this phenomenon. A memory trace of the recent history of network activity is maintained in the state of the synapses, allowing the network to temporarily reactivate the representation of the previous environment in the absence of the corresponding sensory cues. The model predicts that the number of flickering events depends on the amplitude of the ongoing theta rhythm and the distance between the current position of the animal and its position at the time of cue switching. We test these predictions with new analysis of experimental data. These results suggest a potential role of short-term synaptic plasticity in recruiting the activity of different cell assemblies and in shaping hippocampal activity of behaving animals. This article is protected by copyright. All rights reserved.
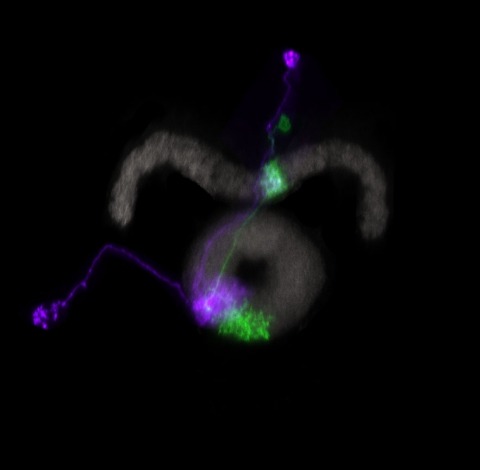
Many animals maintain an internal representation of their heading as they move through their surroundings. Such a compass representation was recently discovered in a neural population in the Drosophila melanogaster central complex, a brain region implicated in spatial navigation. Here, we use two-photon calcium imaging and electrophysiology in head-fixed walking flies to identify a different neural population that conjunctively encodes heading and angular velocity, and is excited selectively by turns in either the clockwise or counterclockwise direction. We show how these mirror-symmetric turn responses combine with the neurons' connectivity to the compass neurons to create an elegant mechanism for updating the fly's heading representation when the animal turns in darkness. This mechanism, which employs recurrent loops with an angular shift, bears a resemblance to those proposed in theoretical models for rodent head direction cells. Our results provide a striking example of structure matching function for a broadly relevant computation.
Ring attractors are a class of recurrent networks hypothesized to underlie the representation of heading direction. Such network structures, schematized as a ring of neurons whose connectivity depends on their heading preferences, can sustain a bump-like activity pattern whose location can be updated by continuous shifts along either turn direction. We recently reported that a population of fly neurons represents the animal's heading via bump-like activity dynamics. We combined two-photon calcium imaging in head-fixed flying flies with optogenetics to overwrite the existing population representation with an artificial one, which was then maintained by the circuit with naturalistic dynamics. A network with local excitation and global inhibition enforces this unique and persistent heading representation. Ring attractor networks have long been invoked in theoretical work; our study provides physiological evidence of their existence and functional architecture.
Place cells in the CA1 region of the hippocampus express location-specific firing despite receiving a steady barrage of heterogeneously tuned excitatory inputs that should compromise output dynamic range and timing. We examined the role of synaptic inhibition in countering the deleterious effects of off-target excitation. Intracellular recordings in behaving mice demonstrate that bimodal excitation drives place cells, while unimodal excitation drives weaker or no spatial tuning in interneurons. Optogenetic hyperpolarization of interneurons had spatially uniform effects on place cell membrane potential dynamics, substantially reducing spatial selectivity. These data and a computational model suggest that spatially uniform inhibitory conductance enhances rate coding in place cells by suppressing out-of-field excitation and by limiting dendritic amplification. Similarly, we observed that inhibitory suppression of phasic noise generated by out-of-field excitation enhances temporal coding by expanding the range of theta phase precession. Thus, spatially uniform inhibition allows proficient and flexible coding in hippocampal CA1 by suppressing heterogeneously tuned excitation.