Mission
To blend the fields of materials chemistry, fluorescence microscopy, and molecular biology in a collaborative process that integrates disparate perspectives, expertise, and methods to advance the field of neuroscience.
Motivation
Among the organs in our body, the brain easily remains the most intriguing in terms of its complexity and function. Nerve cells, which are the functional building blocks of the brain, operate in complex networks that underpin most of the brain’s capabilities, including ability to learn, remember, initiate and orchestrate complex movement. In systems neuroscience, behavioral assays, and large-scale neuronal activity recordings (often aided by biosensors) have broadened our understanding of the role that specific brain regions and neuronal circuits play in a behaving animal. An equally exciting aspect of neuroscience concerns itself with the cellular and molecular underpinnings of nerve cell function. Unique among all cell types, the brain’s constituent cells, neurons, need to communicate with one another in intricate networks that are responsible for circuit function, and most of this communication is enabled by chemical cues that are released between neurons. Better understanding of neuronal communication via chemical release from synapses requires technologies that can help us visualize and measure the spatial and temporal dynamics of these chemical signals. Our lab is interested in developing optical biosensors from nanomaterials that have quantal detection capabilities, high spatial resolution, and excellent signal-to-noise ratio.
Approach
Our lab is part of the Molecular Tools and Imaging (MTI) research area at Janelia. MTI’s mission in Janelia is the development and deployment of tools and technologies that facilitate investigations into various facets of biology. Our lab is interested in making optical biosensors (nanosensors) that can help us visualize the release and diffusion of a class of neurochemicals released from neurons, called neuromodulators (biogenic amines in particular). After developing and validating the tools, we endeavor to address outstanding neurobiology questions that are accessible using our tools but are otherwise elusive when approached with conventional methods of inquiry.
One unique aspect of the work we do is the platform we choose to develop sensors from. While most biosensors that are developed and used in neuroscience are genetically encoded proteins, our lab seeks to get the job done using optical nanomaterials that exhibit a range of advantageous photophysical properties, including photostability and emission in the near infrared region of the spectrum. Furthermore, synthetic (as opposed to protein-based) biosensors offer flexibility from which novel optical tools such as nanofilms, implantable probes can be developed.
Here is an example of a recently developed optical assay from our lab that enables us to visualize the release and diffusion of the neuromodulator dopamine from a single process of a dopamine neuron.
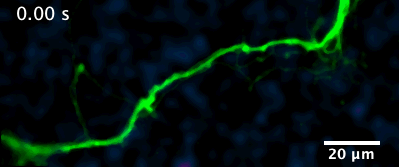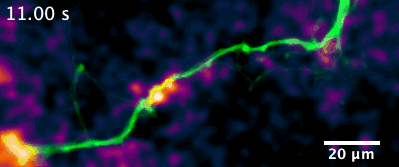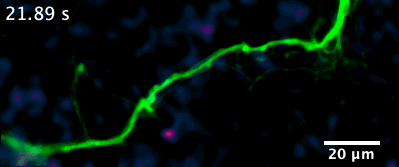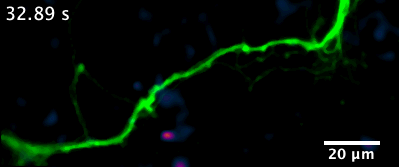
Projects
The lab has several interests that span the fields of neurobiology and materials chemistry.
Neurobiology Projects
The lab has several advanced projects that would benefit from a postdoc with an interest or expertise in molecular and cellular neurobiology of dopaminergic, noradrenergic, and/or serotonergic neurons. You do not need a background in materials, microscopy, chemistry for these projects.
Some of the projects we’re currently working on include:
- Establishing the molecular determinants of release in axon terminals of modulatory neurons (dopaminergic, serotonergic)
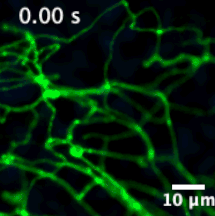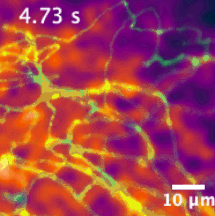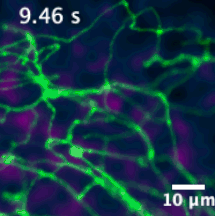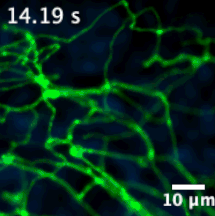
We have developed sensors and microscopes with a unique capability to help us visualize chemical efflux from individual boutons of dopamine neurons, with single release site resolution and quantal sensitivity. This means we can employ post hoc techniques, including immunofluorescence, super resolution imaging, electron microscopy and genetic perturbations to precisely examine the molecular content of each bouton, and map that information onto activity from the bouton.
 |
 |
For example, in this animation (on the left), we can visualize release from many boutons of a dopamine neuron axonal arbor with the resolution of a putative single release site. Each bouton’s molecular signature can then be interrogated post hoc to study the molecular machinery responsible for release. This offers opportunities to answer why a significant population of dopamine varicosities are release incompetent (known from previous literature from our lab and others, see still image on the right). Ultimately, we are interested in uncovering the rules that govern release competency in boutons of modulatory neurons. Using readouts from reduced preparations (culture, brain slices), we then develop hypotheses that can be tested in vivo in model organisms. In that sense, our tools offer us a unique opportunity to bridge the gap between molecular and systems neuroscience.
- Exploring the nature and regulation of dendritic release from modulatory neurons
 |
 |
The animations you see above shows spontaneous release activity from dendritic processes of a dopamine neuron. Dendritic release from modulatory neurons, while not new, is not well understood, primarily because we lack the tools to study these phenomena with high spatial resolution. With the imaging and sensing techniques we’ve developed in the lab, we can now being to characterize the nature of dendritic release with unrivaled precision.
- Exploring the nature and regulation of release from monoamine modulatory neurons
Neurochemical dynamics from dopamine neurons is better understood compared to other biogenic amine neuromodulators such as serotonin and norepinephrine, and neuropeptides. The nature of release from varicosities of serotonergic neurons, for example, remains quite unexplored. We do not know how much of the architecture and properties of release capable boutons in dopamine neurons carries over to these other neuron/neuromodulator types. We have tools that can help us carry out high resolution imaging of these class of modulators and the lab is poised to contribute considerable new knowledge with this line of inquiry.
- Leveraging our tools to study dysfunctions in monoamine neuromodulation
Dopamine, serotonin, and other monoamines are important neuromodulators in the nervous system that play crucial roles in a wide range of physiological functions, including regulating mood, learning, and motor control. Dysregulation of these neurochemicals has been linked to several neurological and psychiatric disorders, including depression, anxiety, and Parkinson's disease. We are interested in studying the molecular basis of neuromodulatory release and, using our assays, take a fresh look at how these go awry in disease states.
Tool Development Projects
The lab is passionately dedicated to developing new tools that can facilitate studies of neuroscience by leveraging its roots and expertise in materials chemistry and nanotechnology. Below are just a few of the projects we’re interested in pursuing:
- Development of high throughput optical assays based on high resolution recording of neurochemical release dynamics
Most of our current optical assays rely on imaging and are relatively low throughput. They are not suitable for carrying out large scale pharmacological screens, CRIPR-based genetic perturbations, single cell sequencing and others that are better suited to addressing several biological questions. We are interested in developing high through assays that can complement our current technologies.
- Development of implantable probes or biocompatible injectable reagents for imaging neuromodulators in vivo
Imaging neurochemical release in vivo at the resolution of a single synapse is currently not possible with any optical technique. We are exploring how our tools can help visualize release in vivo with a better spatial resolution than is currently possible.
- Development of a strategy to develop optical nanosensors quickly and efficiently for a target of interest
There are far more interesting molecules to study than tools to study them with. We are interested in developing biosensors that can help us study other classes of biomolecular targets with the same level of success as we currently do monoamine neuromodulators.
- Investigations into the photophysics and chemistry of near infrared emissive single wall carbon nanotubes and quantum dots
- Developing modern microscopy and plate reader solutions for near infrared and short-wave infrared imaging
Modern fluorescence microscopy relies on silicon-based technology for detecting photons. These detectors (cameras, PMTs) have an efficiency of detection that limits imaging to < 900 nm. Emitters in the near infrared (900 – 1000 nm) and short-wave infrared (1 – 1.7 µm) regions of the spectrum, despite increasingly becoming available for use, are not compatible with most microscopes. Our lab as a standing interesting in bringing modern microscopy solutions to this underutilized (0.9 – 1.7 µm) region of the electromagnetic spectrum.
Techniques
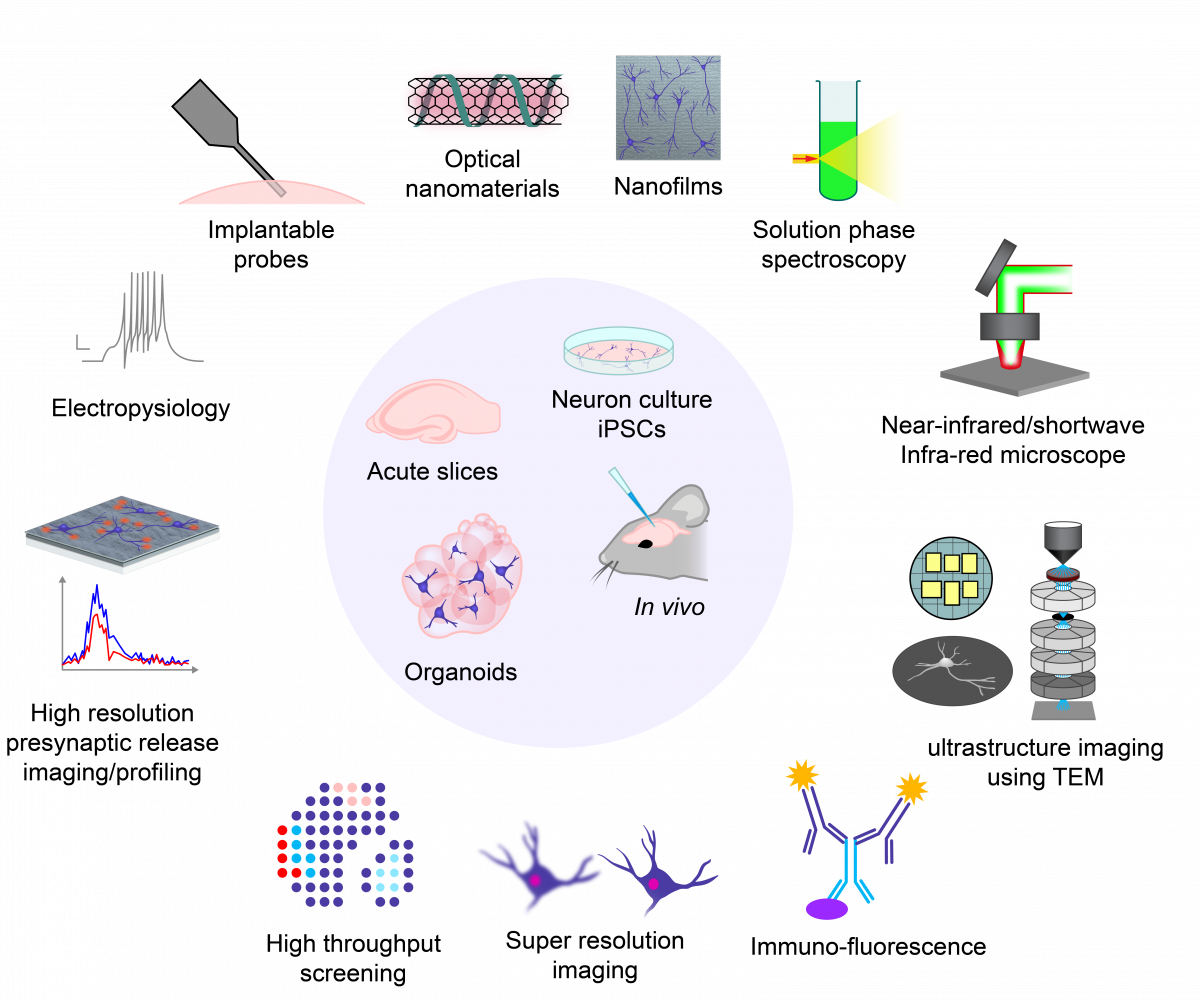
In the lab, you will be exposed to and will have the opportunity to develop expertise in a wide range of laboratory techniques that span the fields of molecular biology, microcopy/imaging/image analysis, ultrastructure imaging using super resolution techniques, electron and expansion microscopy, acute slice and culture electrophysiology and many others. If you’re interested in the materials chemistry aspects of the work we do, you will get additional training in cutting edge nanomaterial synthesis and characterization techniques, tool and technology development. The graphic below is meant to provide a high-level look into various techniques that lie at the intersection of microscopy, nanotechnology, and molecular and cellular neurobiology.
The Lab at a Glance