Towards large-scale neurophysiology based on imaging
Even simple choice behaviors involve large numbers of neurons in multiple brain areas. Due to technical limitations, neurophysiological recordings typically sample only a small subset of neurons, limiting our understanding of neural representations and their relationship to neural circuit structure. Two-photon microscopy has been used to image the activity of populations of individual neurons in behaving animals. Recent advances in genetically encoded calcium indicators (GECIs) permit sensitive detection of neural activity in individual neurons and tracking of activity in neural populations across days or weeks. To date, imaging with cellular resolution in the mammalian brain has been limited to hundreds of neurons in small tissue volumes.
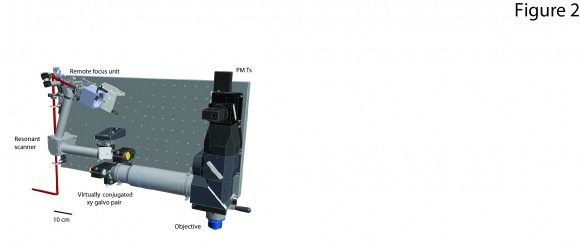
We are developing microscopes and imaging methods to sample very large brain volumes with single neuron resolution. These methods rely on multiple inventions. 1.) Custom microscope optics with very large fields of view, high resolutions, and rapid methods for aberration free 3d scanning (2p-RAM, see sidebar). 3.) Software for instrument control and on-line data analysis. 4.) New ultra-sensitive GECIs. The goal is to break the barrier between analysis at the level of brain areas and single neurons by sampling behavior-related activity in all neurons across multiple brain regions. This work is a collaboration with Dan Flickinger and the GENIE project.
Calcium imaging can also be used to characterize single synaptic inputs on the dendrites of individual neurons. Only tiny subsets of synapses impinging onto single neurons have so far been probed at any given time. Using similar imaging approaches as those highlighted above we also hope to map the receptive fields of a majority of inputs to individual neurons. This will yield a quantitative understanding of how input is converted to spiking output, a key aspect of neural computation. This work is in collaboration with the Ji lab.