Filter
Associated Lab
- Aso Lab (29) Apply Aso Lab filter
- Betzig Lab (1) Apply Betzig Lab filter
- Bock Lab (2) Apply Bock Lab filter
- Branson Lab (7) Apply Branson Lab filter
- Card Lab (5) Apply Card Lab filter
- Clapham Lab (1) Apply Clapham Lab filter
- Dickson Lab (2) Apply Dickson Lab filter
- Druckmann Lab (1) Apply Druckmann Lab filter
- Fetter Lab (1) Apply Fetter Lab filter
- Funke Lab (1) Apply Funke Lab filter
- Harris Lab (3) Apply Harris Lab filter
- Heberlein Lab (2) Apply Heberlein Lab filter
- Hermundstad Lab (2) Apply Hermundstad Lab filter
- Hess Lab (5) Apply Hess Lab filter
- Jayaraman Lab (5) Apply Jayaraman Lab filter
- Lippincott-Schwartz Lab (1) Apply Lippincott-Schwartz Lab filter
- Looger Lab (2) Apply Looger Lab filter
- O'Shea Lab (1) Apply O'Shea Lab filter
- Otopalik Lab (1) Apply Otopalik Lab filter
- Reiser Lab (15) Apply Reiser Lab filter
- Riddiford Lab (1) Apply Riddiford Lab filter
- Romani Lab (1) Apply Romani Lab filter
- Remove Rubin Lab filter Rubin Lab
- Saalfeld Lab (4) Apply Saalfeld Lab filter
- Scheffer Lab (7) Apply Scheffer Lab filter
- Schreiter Lab (1) Apply Schreiter Lab filter
- Simpson Lab (3) Apply Simpson Lab filter
- Singer Lab (1) Apply Singer Lab filter
- Spruston Lab (1) Apply Spruston Lab filter
- Stern Lab (1) Apply Stern Lab filter
- Svoboda Lab (3) Apply Svoboda Lab filter
- Tjian Lab (1) Apply Tjian Lab filter
- Truman Lab (4) Apply Truman Lab filter
- Turaga Lab (1) Apply Turaga Lab filter
- Turner Lab (5) Apply Turner Lab filter
- Zuker Lab (1) Apply Zuker Lab filter
Associated Project Team
Publication Date
- 2024 (3) Apply 2024 filter
- 2023 (6) Apply 2023 filter
- 2022 (1) Apply 2022 filter
- 2021 (4) Apply 2021 filter
- 2020 (9) Apply 2020 filter
- 2019 (6) Apply 2019 filter
- 2018 (7) Apply 2018 filter
- 2017 (15) Apply 2017 filter
- 2016 (3) Apply 2016 filter
- 2015 (16) Apply 2015 filter
- 2014 (9) Apply 2014 filter
- 2013 (5) Apply 2013 filter
- 2012 (8) Apply 2012 filter
- 2011 (4) Apply 2011 filter
- 2010 (4) Apply 2010 filter
- 2009 (2) Apply 2009 filter
- 2008 (4) Apply 2008 filter
- 2007 (2) Apply 2007 filter
- 2006 (1) Apply 2006 filter
- 2002 (1) Apply 2002 filter
- 2000 (2) Apply 2000 filter
- 1999 (1) Apply 1999 filter
- 1997 (1) Apply 1997 filter
- 1995 (2) Apply 1995 filter
- 1994 (2) Apply 1994 filter
- 1993 (2) Apply 1993 filter
- 1992 (1) Apply 1992 filter
- 1991 (2) Apply 1991 filter
- 1990 (3) Apply 1990 filter
- 1989 (2) Apply 1989 filter
- 1987 (2) Apply 1987 filter
- 1986 (1) Apply 1986 filter
- 1985 (1) Apply 1985 filter
- 1984 (1) Apply 1984 filter
- 1983 (1) Apply 1983 filter
- 1982 (2) Apply 1982 filter
- 1981 (1) Apply 1981 filter
- 1979 (1) Apply 1979 filter
- 1973 (1) Apply 1973 filter
Type of Publication
139 Publications
Showing 61-70 of 139 resultsThe connectome provides large scale connectivity and morphology information for the majority of the central brain of . Using this data set, we provide a complete description of the olfactory system, covering all first, second and lateral horn-associated third-order neurons. We develop a generally applicable strategy to extract information flow and layered organisation from connectome graphs, mapping olfactory input to descending interneurons. This identifies a range of motifs including highly lateralised circuits in the antennal lobe and patterns of convergence downstream of the mushroom body and lateral horn. Leveraging a second data set we provide a first quantitative assessment of inter- versus intra-individual stereotypy. Comparing neurons across two brains (three hemispheres) reveals striking similarity in neuronal morphology across brains. Connectivity correlates with morphology and neurons of the same morphological type show similar connection variability within the same brain as across two brains.
Different types of Drosophila dopaminergic neurons (DANs) reinforce memories of unique valence and provide state-dependent motivational control [1]. Prior studies suggest that the compartment architecture of the mushroom body (MB) is the relevant resolution for distinct DAN functions [2, 3]. Here we used a recent electron microscope volume of the fly brain [4] to reconstruct the fine anatomy of individual DANs within three MB compartments. We find the 20 DANs of the γ5 compartment, at least some of which provide reward teaching signals, can be clustered into 5 anatomical subtypes that innervate different regions within γ5. Reconstructing 821 upstream neurons reveals input selectivity, supporting the functional relevance of DAN sub-classification. Only one PAM-γ5 DAN subtype (γ5fb) receives direct recurrent input from γ5β’2a mushroom body output neurons (MBONs) and behavioral experiments distinguish a role for these DANs in memory revaluation from those reinforcing sugar memory. Other DAN subtypes receive major, and potentially reinforcing, inputs from putative gustatory interneurons or lateral horn neurons, which can also relay indirect feedback from the MB. We similarly reconstructed the single aversively reinforcing PPL1-γ1pedc DAN. The γ1pedc DAN inputs are mostly different to those of γ5 DANs and are clustered onto distinct branches of its dendritic tree, presumably separating its established roles in aversive reinforcement and appetitive motivation [5, 6]. Additional tracing identified neurons that provide broad input to γ5, β’2a and γ1pedc DANs suggesting that distributed DAN populations can be coordinately regulated. These connectomic and behavioral analyses therefore reveal additional complexity of dopaminergic reinforcement circuits between and within MB compartments.
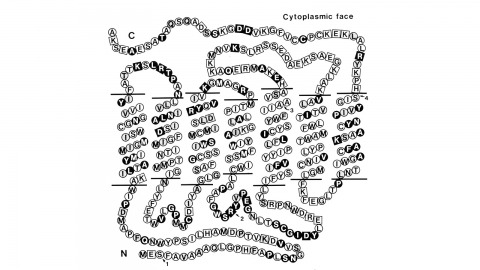
Using a novel method for detecting cross-homologous nucleic acid sequences we have isolated the gene coding for the major rhodopsin of Drosophila melanogaster and mapped it to chromosomal region 92B8-11. Comparison of cDNA and genomic DNA sequences indicates that the gene is divided into five exons. The amino acid sequence deduced from the nucleotide sequence is 373 residues long, and the polypeptide chain contains seven hydrophobic segments that appear to correspond to the seven transmembrane segments characteristic of other rhodopsins. Three regions of Drosophila rhodopsin are highly conserved with the corresponding domains of bovine rhodopsin, suggesting an important role for these polypeptide regions.
Janelia Farm, the new research campus of the Howard Hughes Medical Institute, is an ongoing experiment in the social engineering of research communities.
The development of the adult optic lobe (OL) of is directed by a wave of ingrowth of the photoreceptors over a two day period at the outset of metamorphosis which is accompanied by the appearance of the pupal-specific transcription factor Broad-Z3 (Br-Z3) and expression of early drivers in OL neurons. During this time, there are pulses of ecdysteroids that time the metamorphic events. At the outset, the transient appearance of juvenile hormone (JH) prevents precocious development of the OL caused by the ecdysteroid peak that initiates pupariation, but the artificial maintenance of JH after this time misdirects subsequent development. Axon ingrowth, Br-Z3 appearance and the expression of early drivers were unaffected, but aspects of later development such as the dendritic expansion of the lamina monopolar neurons and the expression of late drivers were suppressed. This effect of the exogenous JH mimic (JHM) pyriproxifen is lost by 24 hr after pupariation. Part of this effect of JHM is due to its suppression of the appearance of ecdysone receptor EcR-B1 that occurs after pupation and during early adult development.
The Drosophila gene sevenless encodes a putative trans-membrane receptor required for the formation of one particular cell, the R7 photoreceptor, in each ommatidium of the compound eye. Mutations in this gene result in the cell normally destined to form the R7 cell forming a non-neuronal cell type instead. These observations have led to the proposal that the sevenless protein receives at least part of the positional information required for the R7 developmental pathway. We have generated antibodies specific for sevenless and have examined expression of the protein by light and electron microscopy. sevenless protein is present transiently at high levels in at least 9 cells in each developing ommatidium and is detectable several hours before any overt differentiation of R7. The protein is mostly localized at the apices of the cells, in microvilli, but is also found deeper in the tissue where certain cells contact the R8 cell. This finding suggests that R8 expresses a ligand for the sevenless protein.
Several aspects of locomotor control have been ascribed to the central complex of the insect brain; however, the role of distinct substructures of this complex is not well known. The tay bridge1 (tay1) mutant of Drosophila melanogaster was originally isolated on the basis of reduced walking speed and activity. In addition, tay1 is defective in the compensation of rotatory stimuli during walking and histologically, tay1 causes a mid-sagittal constriction of the protocerebral bridge, a constituent of the central complex. Cloning of the tay gene revealed that it encodes a novel protein with no significant homology to any known protein. To associate the behavioral phenotypes with the anatomical defect in the protocerebral bridge, we used different driver lines to express the tay cDNA in various neuronal subpopulations of the central brain in tay1-mutant flies. These experiments showed an association of the aberrant walking speed and activity with the structural defect in the protocerebral bridge. In contrast, the compensation of rotatory stimuli during walking was rescued without a restoration of the protocerebral bridge. The results of our differential rescue approach are supported by neuronal silencing experiments using conditional tetanus toxin expression in the same subset of neurons. These findings show for the first time that the walking speed and activity is controlled by different substructures of the central brain than the compensatory locomotion for rotatory stimuli.
Starting a new research campus is a leap of faith. Only later, in the full measure of time, is it possible to take stock of what has worked and what could have been done better or differently. The Janelia Research Campus opened its doors 12 years ago. What has it achieved? What has it taught us? And where does Janelia go from here?
The Drosophila cerebrum originates from about 100 neuroblasts per hemisphere, with each neuroblast producing a characteristic set of neurons. Neurons from a neuroblast are often so diverse that many neuron types remain unexplored. We developed new genetic tools that target neuroblasts and their diverse descendants, increasing our ability to study fly brain structure and development. Common enhancer-based drivers label neurons on the basis of terminal identities rather than origins, which provides limited labeling in the heterogeneous neuronal lineages. We successfully converted conventional drivers that are temporarily expressed in neuroblasts, into drivers expressed in all subsequent neuroblast progeny. One technique involves immortalizing GAL4 expression in neuroblasts and their descendants. Another depends on loss of the GAL4 repressor, GAL80, from neuroblasts during early neurogenesis. Furthermore, we expanded the diversity of MARCM-based reagents and established another site-specific mitotic recombination system. Our transgenic tools can be combined to map individual neurons in specific lineages of various genotypes.
Identifying the neurotransmitters used by specific neurons is a critical step in understanding the function of neural circuits. However, methods for the consistent and efficient detection of neurotransmitter markers remain limited. Fluorescence hybridization (FISH) enables direct labeling of type-specific mRNA in neurons. Recent advances in FISH allow this technique to be carried out in intact tissue samples such as whole-mount brains. Here, we present a FISH platform for high-throughput detection of eight common neurotransmitter phenotypes in brains. We greatly increase FISH throughput by processing samples mounted on coverslips and optimizing fluorophore choice for each probe to facilitate multiplexing. As application examples, we demonstrate cases of neurotransmitter co-expression, reveal neurotransmitter phenotypes of specific cell types and explore the onset of neurotransmitter expression in the developing optic lobe. Beyond neurotransmitter markers, our protocols can in principle be used for large scale FISH detection of any mRNA in whole-mount fly brains.