Filter
Associated Lab
- Ahrens Lab (4) Apply Ahrens Lab filter
- Betzig Lab (1) Apply Betzig Lab filter
- Druckmann Lab (1) Apply Druckmann Lab filter
- Harris Lab (4) Apply Harris Lab filter
- Hermundstad Lab (1) Apply Hermundstad Lab filter
- Jayaraman Lab (9) Apply Jayaraman Lab filter
- Karpova Lab (1) Apply Karpova Lab filter
- Lavis Lab (5) Apply Lavis Lab filter
- Leonardo Lab (1) Apply Leonardo Lab filter
- Liu (Zhe) Lab (1) Apply Liu (Zhe) Lab filter
- Looger Lab (24) Apply Looger Lab filter
- Podgorski Lab (5) Apply Podgorski Lab filter
- Rubin Lab (1) Apply Rubin Lab filter
- Remove Schreiter Lab filter Schreiter Lab
- Svoboda Lab (13) Apply Svoboda Lab filter
- Tillberg Lab (1) Apply Tillberg Lab filter
- Turner Lab (3) Apply Turner Lab filter
- Zlatic Lab (1) Apply Zlatic Lab filter
Associated Project Team
Publication Date
- 2024 (2) Apply 2024 filter
- 2023 (6) Apply 2023 filter
- 2021 (1) Apply 2021 filter
- 2020 (5) Apply 2020 filter
- 2019 (4) Apply 2019 filter
- 2018 (4) Apply 2018 filter
- 2017 (4) Apply 2017 filter
- 2016 (2) Apply 2016 filter
- 2015 (4) Apply 2015 filter
- 2013 (7) Apply 2013 filter
- 2012 (2) Apply 2012 filter
- 2011 (3) Apply 2011 filter
- 2010 (2) Apply 2010 filter
- 2009 (3) Apply 2009 filter
- 2008 (3) Apply 2008 filter
- 2007 (3) Apply 2007 filter
- 2006 (2) Apply 2006 filter
- 2003 (1) Apply 2003 filter
- 2001 (2) Apply 2001 filter
- 1999 (1) Apply 1999 filter
Type of Publication
61 Publications
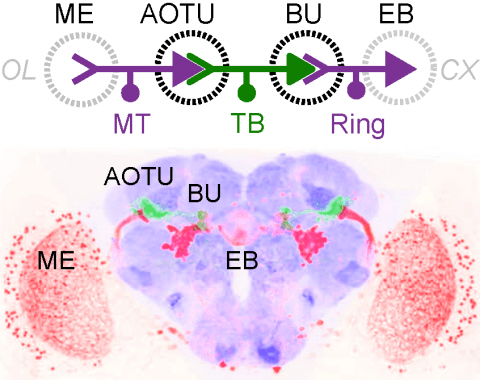
Showing 41-50 of 61 resultsMany animals orient using visual cues, but how a single cue is selected from among many is poorly understood. Here we show that Drosophila ring neurons—central brain neurons implicated in navigation—display visual stimulus selection. Using in vivo two-color two-photon imaging with genetically encoded calcium indicators, we demonstrate that individual ring neurons inherit simple-cell-like receptive fields from their upstream partners. Stimuli in the contralateral visual field suppressed responses to ipsilateral stimuli in both populations. Suppression strength depended on when and where the contralateral stimulus was presented, an effect stronger in ring neurons than in their upstream inputs. This history-dependent effect on the temporal structure of visual responses, which was well modeled by a simple biphasic filter, may determine how visual references are selected for the fly's internal compass. Our approach highlights how two-color calcium imaging can help identify and localize the origins of sensory transformations across synaptically connected neural populations.
Metal ion homeostasis is critical to the survival of all cells. Regulation of nickel concentrations in Escherichia coli is mediated by the NikR repressor via nickel-induced transcriptional repression of the nickel ABC-type transporter, NikABCDE. Here, we report two crystal structures of nickel-activated E. coli NikR, the isolated repressor at 2.1 A resolution and in a complex with its operator DNA sequence from the nik promoter at 3.1 A resolution. Along with the previously published structure of apo-NikR, these structures allow us to evaluate functional proposals for how metal ions activate NikR, delineate the drastic conformational changes required for operator recognition, and describe the formation of a second metal-binding site in the presence of DNA. They also provide a rare set of structural views of a ligand-responsive transcription factor in the unbound, ligand-induced, and DNA-bound states, establishing a model system for the study of ligand-mediated effects on transcription factor function.
Genetically encoded calcium indicators (GECIs) are powerful tools for systems neuroscience. Recent efforts in protein engineering have significantly increased the performance of GECIs. The state-of-the art single-wavelength GECI, GCaMP3, has been deployed in a number of model organisms and can reliably detect three or more action potentials in short bursts in several systems in vivo . Through protein structure determination, targeted mutagenesis, high-throughput screening, and a battery of in vitro assays, we have increased the dynamic range of GCaMP3 by severalfold, creating a family of “GCaMP5” sensors. We tested GCaMP5s in several systems: cultured neurons and astrocytes, mouse retina, and in vivo in Caenorhabditis chemosensory neurons, Drosophila larval neuromuscular junction and adult antennal lobe, zebrafish retina and tectum, and mouse visual cortex. Signal-to-noise ratio was improved by at least 2- to 3-fold. In the visual cortex, two GCaMP5 variants detected twice as many visual stimulus-responsive cells as GCaMP3. By combining in vivo imaging with electrophysiology we show that GCaMP5 fluorescence provides a more reliable measure of neuronal activity than its predecessor GCaMP3.GCaMP5allows more sensitive detection of neural activity in vivo andmayfind widespread applications for cellular imaging in general.
Genetically encoded pH sensors based on fluorescent proteins are valuable tools for the imaging of cellular events that are associated with pH changes, such as exocytosis and endocytosis. Superecliptic pHluorin (SEP) is a pH-sensitive green fluorescent protein (GFP) variant widely used for such applications. Here, we report the rational design, development, structure, and applications of Lime, an improved SEP variant with higher fluorescence brightness and greater pH sensitivity. The X-ray crystal structure of Lime supports the mechanistic rationale that guided the introduction of beneficial mutations. Lime provides substantial improvements relative to SEP for imaging of endocytosis and exocytosis. Furthermore, Lime and its variants are advantageous for a broader range of applications including the detection of synaptic release and neuronal voltage changes.
The ribbon-helix-helix (RHH) superfamily of transcription factors uses a conserved three-dimensional structural motif to bind to DNA in a sequence-specific manner. This functionally diverse protein superfamily regulates the transcription of genes that are involved in the uptake of metals, amino-acid biosynthesis, cell division, the control of plasmid copy number, the lytic cycle of bacteriophages and, perhaps, many other cellular processes. In this Analysis, the structures of different RHH transcription factors are compared in order to evaluate the sequence motifs that are required for RHH-domain folding and DNA binding, as well as to identify conserved protein-DNA interactions in this superfamily.
The thioredoxin system helps maintain a reducing environment in cells, but thioredoxin functions as more than simply an antioxidant. Thioredoxin functions depend on the protein's redox state, as determined by two conserved cysteines. Key biologic activities of thioredoxin include antioxidant, growth control, and antiapoptotic properties, resulting from interaction with target molecules including transcription factors. Mechanisms by which thioredoxin regulates cell growth include binding to signaling molecules such as apoptosis signal-regulating kinase-1 (ASK-1) and thioredoxin-interacting protein (Txnip). The molecular interplay between thioredoxin, ASK-1, and Txnip potentially influences cell growth and survival in diverse human diseases such as cancer, diabetes, and heart disease. In this review, we focus on the structure of thioredoxin and its functional regulation of cell growth through the interactions with signaling molecules.
S-nitrosylation is a post-translational protein modification that can alter the function of a variety of proteins. Despite the growing wealth of information that this modification may have important functional consequences, little is known about the structure of the moiety or its effect on protein tertiary structure. Here we report high-resolution x-ray crystal structures of S-nitrosylated and unmodified blackfin tuna myoglobin, which demonstrate that in vitro S-nitrosylation of this protein at the surface-exposed Cys-10 directly causes a reversible conformational change by "wedging" apart a helix and loop. Furthermore, we have demonstrated in solution and in a single crystal that reduction of the S-nitrosylated myoglobin with dithionite results in NO cleavage from the sulfur of Cys-10 and rebinding to the reduced heme iron, showing the reversibility of both the modification and the conformational changes. Finally, we report the 0.95-A structure of ferrous nitrosyl myoglobin, which provides an accurate structural view of the NO coordination geometry in the context of a globin heme pocket.
Genetically encoded calcium indicators (GECIs) allow measurement of activity in large populations of neurons and in small neuronal compartments, over times of milliseconds to months. Although GFP-based GECIs are widely used for in vivo neurophysiology, GECIs with red-shifted excitation and emission spectra have advantages for in vivo imaging because of reduced scattering and absorption in tissue, and a consequent reduction in phototoxicity. However, current red GECIs are inferior to the state-of-the-art GFP-based GCaMP6 indicators for detecting and quantifying neural activity. Here we present improved red GECIs based on mRuby (jRCaMP1a, b) and mApple (jRGECO1a), with sensitivity comparable to GCaMP6. We characterized the performance of the new red GECIs in cultured neurons and in mouse, Drosophila, zebrafish and C. elegans in vivo. Red GECIs facilitate deep-tissue imaging, dual-color imaging together with GFP-based reporters, and the use of optogenetics in combination with calcium imaging.
The ability to optically image cellular transmembrane voltages at millisecond-timescale resolutions can offer unprecedented insight into the function of living brains in behaving animals. Here, we present a point mutation that increases the sensitivity of Ace2 opsin-based voltage indicators. We use the mutation to develop Voltron2, an improved chemigeneic voltage indicator that has a 65% higher sensitivity to single APs and 3-fold higher sensitivity to subthreshold potentials than Voltron. Voltron2 retained the sub-millisecond kinetics and photostability of its predecessor, although with lower baseline fluorescence. In multiple in vitro and in vivo comparisons with its predecessor across multiple species, we found Voltron2 to be more sensitive to APs and subthreshold fluctuations. Finally, we used Voltron2 to study and evaluate the possible mechanisms of interneuron synchronization in the mouse hippocampus. Overall, we have discovered a generalizable mutation that significantly increases the sensitivity of Ace2 rhodopsin-based sensors, improving their voltage reporting capability.
The phenotypical severity of sickle cell disease (SCD) can be mitigated by modifying mutant hemoglobin S (Hb S, Hb α2β2s) to contain embryonic ζ globin in place of adult α-globin subunits (Hb ζ2β2s). Crystallographical analyses of liganded Hb ζζ2β2s, though, demonstrate a tense (T-state) quaternary structure that paradoxically predicts its participation in--rather than its exclusion from--pathological deoxyHb S polymers. We resolved this structure-function conundrum by examining the effects of α → ζ exchange on the characteristics of specific amino acids that mediate sickle polymer assembly. Superposition analyses of the βs subunits of T-state deoxyHb α2β2s and T-state CO-liganded Hb ζ2β2s reveal significant displacements of both mutant βsVal6 and conserved β-chain contact residues, predicting weakening of corresponding polymer-stabilizing interactions. Similar comparisons of the α- and ζ-globin subunits implicate four amino acids that are either repositioned or undergo non-conservative substitution, abrogating critical polymer contacts. CO-Hb ζ2βs2 additionally exhibits a unique trimer-of-heterotetramers crystal packing that is sustained by novel intermolecular interactions involving the pathological βsVal6, contrasting sharply with the classical double-stranded packing of deoxyHb S. Finally, the unusually large buried solvent-accessible surface area for CO-Hb ζ2β2s suggests that it does not co-assemble with deoxyHb S in vivo . In sum, the antipolymer activities of Hb ζ2β2s appear to arise from both repositioning and replacement of specific α- and βs-chain residues, favoring an alternate T-state solution structure that is excluded from pathological deoxyHb S polymers. These data account for the antipolymer activity of Hb ζ2β2s, and recommend the utility of SCD therapeutics that capitalize on α-globin exchange strategies.