Filter
Associated Lab
- Aguilera Castrejon Lab (1) Apply Aguilera Castrejon Lab filter
- Aso Lab (3) Apply Aso Lab filter
- Betzig Lab (1) Apply Betzig Lab filter
- Bock Lab (2) Apply Bock Lab filter
- Branson Lab (3) Apply Branson Lab filter
- Card Lab (1) Apply Card Lab filter
- Cardona Lab (4) Apply Cardona Lab filter
- Clapham Lab (1) Apply Clapham Lab filter
- Cui Lab (1) Apply Cui Lab filter
- Dickson Lab (1) Apply Dickson Lab filter
- Druckmann Lab (2) Apply Druckmann Lab filter
- Eddy/Rivas Lab (2) Apply Eddy/Rivas Lab filter
- Egnor Lab (1) Apply Egnor Lab filter
- Fetter Lab (3) Apply Fetter Lab filter
- Freeman Lab (1) Apply Freeman Lab filter
- Funke Lab (1) Apply Funke Lab filter
- Gonen Lab (1) Apply Gonen Lab filter
- Harris Lab (3) Apply Harris Lab filter
- Heberlein Lab (1) Apply Heberlein Lab filter
- Hess Lab (6) Apply Hess Lab filter
- Jayaraman Lab (3) Apply Jayaraman Lab filter
- Keller Lab (1) Apply Keller Lab filter
- Lavis Lab (1) Apply Lavis Lab filter
- Lee (Albert) Lab (1) Apply Lee (Albert) Lab filter
- Li Lab (1) Apply Li Lab filter
- Lippincott-Schwartz Lab (2) Apply Lippincott-Schwartz Lab filter
- Liu (Zhe) Lab (1) Apply Liu (Zhe) Lab filter
- Looger Lab (1) Apply Looger Lab filter
- Magee Lab (1) Apply Magee Lab filter
- Otopalik Lab (1) Apply Otopalik Lab filter
- Reiser Lab (1) Apply Reiser Lab filter
- Rubin Lab (9) Apply Rubin Lab filter
- Saalfeld Lab (7) Apply Saalfeld Lab filter
- Satou Lab (1) Apply Satou Lab filter
- Scheffer Lab (7) Apply Scheffer Lab filter
- Singer Lab (1) Apply Singer Lab filter
- Spruston Lab (2) Apply Spruston Lab filter
- Stern Lab (2) Apply Stern Lab filter
- Sternson Lab (1) Apply Sternson Lab filter
- Stringer Lab (1) Apply Stringer Lab filter
- Svoboda Lab (2) Apply Svoboda Lab filter
- Tillberg Lab (1) Apply Tillberg Lab filter
- Truman Lab (1) Apply Truman Lab filter
- Turaga Lab (1) Apply Turaga Lab filter
- Turner Lab (2) Apply Turner Lab filter
- Zlatic Lab (1) Apply Zlatic Lab filter
Associated Project Team
Associated Support Team
- Anatomy and Histology (1) Apply Anatomy and Histology filter
- Fly Facility (3) Apply Fly Facility filter
- Gene Targeting and Transgenics (1) Apply Gene Targeting and Transgenics filter
- Janelia Experimental Technology (3) Apply Janelia Experimental Technology filter
- Management Team (1) Apply Management Team filter
- Project Technical Resources (5) Apply Project Technical Resources filter
- Quantitative Genomics (1) Apply Quantitative Genomics filter
- Remove Scientific Computing Software filter Scientific Computing Software
- Scientific Computing Systems (1) Apply Scientific Computing Systems filter
Publication Date
59 Janelia Publications
Showing 51-59 of 59 resultsWe reconstructed the synaptic circuits of seven columns in the second neuropil or medulla behind the fly's compound eye. These neurons embody some of the most stereotyped circuits in one of the most miniaturized of animal brains. The reconstructions allow us, for the first time to our knowledge, to study variations between circuits in the medulla's neighboring columns. This variation in the number of synapses and the types of their synaptic partners has previously been little addressed because methods that visualize multiple circuits have not resolved detailed connections, and existing connectomic studies, which can see such connections, have not so far examined multiple reconstructions of the same circuit. Here, we address the omission by comparing the circuits common to all seven columns to assess variation in their connection strengths and the resultant rates of several different and distinct types of connection error. Error rates reveal that, overall, <1% of contacts are not part of a consensus circuit, and we classify those contacts that supplement (E+) or are missing from it (E-). Autapses, in which the same cell is both presynaptic and postsynaptic at the same synapse, are occasionally seen; two cells in particular, Dm9 and Mi1, form ≥20-fold more autapses than do other neurons. These results delimit the accuracy of developmental events that establish and normally maintain synaptic circuits with such precision, and thereby address the operation of such circuits. They also establish a precedent for error rates that will be required in the new science of connectomics.
Understanding the cell-type composition and spatial organization of brain regions is crucial for interpreting brain computation and function. In the thalamus, the anterior thalamic nuclei (ATN) are involved in a wide variety of functions, yet the cell-type composition of the ATN remains unmapped at a single-cell and spatial resolution. Combining single-cell RNA sequencing, spatial transcriptomics, and multiplexed fluorescent in situ hybridization, we identify three discrete excitatory cell-type clusters that correspond to the known nuclei of the ATN and uncover marker genes, molecular pathways, and putative functions of these cell types. We further illustrate graded spatial variation along the dorsomedial-ventrolateral axis for all individual nuclei of the ATN and additionally demonstrate that the anteroventral nucleus exhibits spatially covarying protein products and long-range inputs. Collectively, our study reveals discrete and continuous cell-type organizational principles of the ATN, which will help to guide and interpret experiments on ATN computation and function.
The formation of large, well-ordered crystals for crystallographic experiments remains a crucial bottleneck to the structural understanding of many important biological systems. To help alleviate this problem in crystallography, we have developed the MicroED method for the collection of electron diffraction data from 3D microcrystals and nanocrystals of radiation-sensitive biological material. In this approach, liquid solutions containing protein microcrystals are deposited on carbon-coated electron microscopy grids and are vitrified by plunging them into liquid ethane. MicroED data are collected for each selected crystal using cryo-electron microscopy, in which the crystal is diffracted using very few electrons as the stage is continuously rotated. This protocol gives advice on how to identify microcrystals by light microscopy or by negative-stain electron microscopy in samples obtained from standard protein crystallization experiments. The protocol also includes information about custom-designed equipment for controlling crystal rotation and software for recording experimental parameters in diffraction image metadata. Identifying microcrystals, preparing samples and setting up the microscope for diffraction data collection take approximately half an hour for each step. Screening microcrystals for quality diffraction takes roughly an hour, and the collection of a single data set is ∼10 min in duration. Complete data sets and resulting high-resolution structures can be obtained from a single crystal or by merging data from multiple crystals.
Repetitive DNA, especially that due to transposable elements (TEs), makes up a large fraction of many genomes. Dfam is an open access database of families of repetitive DNA elements, in which each family is represented by a multiple sequence alignment and a profile hidden Markov model (HMM). The initial release of Dfam, featured in the 2013 NAR Database Issue, contained 1143 families of repetitive elements found in humans, and was used to produce more than 100 Mb of additional annotation of TE-derived regions in the human genome, with improved speed. Here, we describe recent advances, most notably expansion to 4150 total families including a comprehensive set of known repeat families from four new organisms (mouse, zebrafish, fly and nematode). We describe improvements to coverage, and to our methods for identifying and reducing false annotation. We also describe updates to the website interface. The Dfam website has moved to http://dfam.org. Seed alignments, profile HMMs, hit lists and other underlying data are available for download.
Drosophila melanogaster plays an important role in molecular, genetic, and genomic studies of heredity, development, metabolism, behavior, and human disease. The initial reference genome sequence reported more than a decade ago had a profound impact on progress in Drosophila research, and improving the accuracy and completeness of this sequence continues to be important to further progress. We previously described improvement of the 117-Mb sequence in the euchromatic portion of the genome and 21 Mb in the heterochromatic portion, using a whole-genome shotgun assembly, BAC physical mapping, and clone-based finishing. Here, we report an improved reference sequence of the single-copy and middle-repetitive regions of the genome, produced using cytogenetic mapping to mitotic and polytene chromosomes, clone-based finishing and BAC fingerprint verification, ordering of scaffolds by alignment to cDNA sequences, incorporation of other map and sequence data, and validation by whole-genome optical restriction mapping. These data substantially improve the accuracy and completeness of the reference sequence and the order and orientation of sequence scaffolds into chromosome arm assemblies. Representation of the Y chromosome and other heterochromatic regions is particularly improved. The new 143.9-Mb reference sequence, designated Release 6, effectively exhausts clone-based technologies for mapping and sequencing. Highly repeat-rich regions, including large satellite blocks and functional elements such as the ribosomal RNA genes and the centromeres, are largely inaccessible to current sequencing and assembly methods and remain poorly represented. Further significant improvements will require sequencing technologies that do not depend on molecular cloning and that produce very long reads.
In the hippocampus, the classical pyramidal cell type of the subiculum acts as a primary output, conveying hippocampal signals to a diverse suite of downstream regions. Accumulating evidence suggests that the subiculum pyramidal cell population may actually be comprised of discrete subclasses. Here, we investigated the extent and organizational principles governing pyramidal cell heterogeneity throughout the mouse subiculum. Using single-cell RNA-seq, we find that the subiculum pyramidal cell population can be deconstructed into eight separable subclasses. These subclasses were mapped onto abutting spatial domains, ultimately producing a complex laminar and columnar organization with heterogeneity across classical dorsal-ventral, proximal-distal, and superficial-deep axes. We further show that these transcriptomically defined subclasses correspond to differential protein products and can be associated with specific projection targets. This work deconstructs the complex landscape of subiculum pyramidal cells into spatially segregated subclasses that may be observed, controlled, and interpreted in future experiments.
Insects and mammals share similarities of neural organization underlying the perception of odors, taste, vision, sound, and gravity. We observed that insect somatosensation also corresponds to that of mammals. In Drosophila, the projections of all the somatosensory neuron types to the insect's equivalent of the spinal cord segregated into modality-specific layers comparable to those in mammals. Some sensory neurons innervate the ventral brain directly to form modality-specific and topological somatosensory maps. Ascending interneurons with dendrites in matching layers of the nerve cord send axons that converge to respective brain regions. Pathways arising from leg somatosensory neurons encode distinct qualities of leg movement information and play different roles in ground detection. Establishment of the ground pattern and genetic tools for neuronal manipulation should provide the basis for elucidating the mechanisms underlying somatosensation.
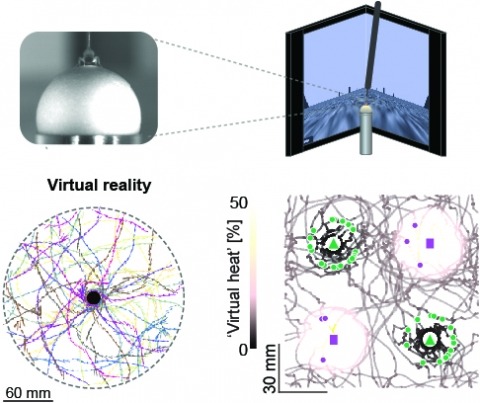
Studying the intertwined roles of sensation, experience, and directed action in navigation has been facilitated by the development of virtual reality (VR) environments for head-fixed animals, allowing for quantitative measurements of behavior in well-controlled conditions. VR has long featured in studies of Drosophila melanogaster, but these experiments have typically allowed the fly to change only its heading in a visual scene and not its position. Here we explore how flies move in two dimensions (2D) using a visual VR environment that more closely captures an animal's experience during free behavior. We show that flies' 2D interaction with landmarks cannot be automatically derived from their orienting behavior under simpler one-dimensional (1D) conditions. Using novel paradigms, we then demonstrate that flies in 2D VR adapt their behavior in response to optogenetically delivered appetitive and aversive stimuli. Much like free-walking flies after encounters with food, head-fixed flies exploring a 2D VR respond to optogenetic activation of sugar-sensing neurons by initiating a local search, which appears not to rely on visual landmarks. Visual landmarks can, however, help flies to avoid areas in VR where they experience an aversive, optogenetically generated heat stimulus. By coupling aversive virtual heat to the flies' presence near visual landmarks of specific shapes, we elicit selective learned avoidance of those landmarks. Thus, we demonstrate that head-fixed flies adaptively navigate in 2D virtual environments, but their reliance on visual landmarks is context dependent. These behavioral paradigms set the stage for interrogation of the fly brain circuitry underlying flexible navigation in complex multisensory environments.
Cells contain hundreds of organelles and macromolecular assemblies. Obtaining a complete understanding of their intricate organization requires the nanometre-level, three-dimensional reconstruction of whole cells, which is only feasible with robust and scalable automatic methods. Here, to support the development of such methods, we annotated up to 35 different cellular organelle classes-ranging from endoplasmic reticulum to microtubules to ribosomes-in diverse sample volumes from multiple cell types imaged at a near-isotropic resolution of 4 nm per voxel with focused ion beam scanning electron microscopy (FIB-SEM). We trained deep learning architectures to segment these structures in 4 nm and 8 nm per voxel FIB-SEM volumes, validated their performance and showed that automatic reconstructions can be used to directly quantify previously inaccessible metrics including spatial interactions between cellular components. We also show that such reconstructions can be used to automatically register light and electron microscopy images for correlative studies. We have created an open data and open-source web repository, 'OpenOrganelle', to share the data, computer code and trained models, which will enable scientists everywhere to query and further improve automatic reconstruction of these datasets.