Filter
Associated Lab
4 Janelia Publications
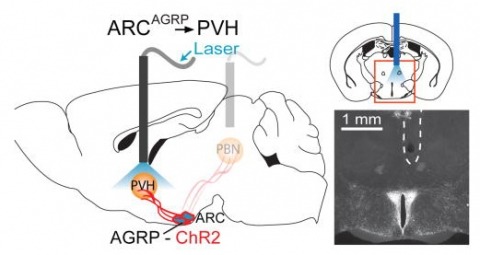
Showing 1-4 of 4 resultsHunger is a complex behavioural state that elicits intense food seeking and consumption. These behaviours are rapidly recapitulated by activation of starvation-sensitive AGRP neurons, which present an entry point for reverse-engineering neural circuits for hunger. Here we mapped synaptic interactions of AGRP neurons with multiple cell populations in mice and probed the contribution of these distinct circuits to feeding behaviour using optogenetic and pharmacogenetic techniques. An inhibitory circuit with paraventricular hypothalamus (PVH) neurons substantially accounted for acute AGRP neuron-evoked eating, whereas two other prominent circuits were insufficient. Within the PVH, we found that AGRP neurons target and inhibit oxytocin neurons, a small population that is selectively lost in Prader-Willi syndrome, a condition involving insatiable hunger. By developing strategies for evaluating molecularly defined circuits, we show that AGRP neuron suppression of oxytocin neurons is critical for evoked feeding. These experiments reveal a new neural circuit that regulates hunger state and pathways associated with overeating disorders.
Mice lacking leptin receptors are grossly obese and diabetic, in part due to dysfunction in brain circuits important for energy homeostasis. Transplantation of leptin receptor-expressing hypothalamic progenitor neurons into the brains of leptin receptor deficient mice led to integration into neural circuits, reduced obesity, and normalized circulating glucose levels.
Transforming synaptic input into action potential output is a fundamental function of neurons. The pattern of action potential output from principal cells of the mammalian hippocampus encodes spatial and nonspatial information, but the cellular and circuit mechanisms by which neurons transform their synaptic input into a given output are unknown. Using a combination of optical activation and cell type-specific pharmacogenetic silencing in vitro, we found that dendritic inhibition is the primary regulator of input-output transformations in mouse hippocampal CA1 pyramidal cells, and acts by gating the dendritic electrogenesis driving burst spiking. Dendrite-targeting interneurons are themselves modulated by interneurons targeting pyramidal cell somata, providing a synaptic substrate for tuning pyramidal cell output through interactions in the local inhibitory network. These results provide evidence for a division of labor in cortical circuits, where distinct computational functions are implemented by subtypes of local inhibitory neurons.
Small molecules are important tools to measure and modulate intracellular signaling pathways. A longstanding limitation for using chemical compounds in complex tissues has been the inability to target bioactive small molecules to a specific cell class. Here, we describe a generalizable esterase-ester pair capable of targeted delivery of small molecules to living cells and tissue with cellular specificity. We used fluorogenic molecules to rapidly identify a small ester masking motif that is stable to endogenous esterases, but is efficiently removed by an exogenous esterase. This strategy allows facile targeting of dyes and drugs in complex biological environments to label specific cell types, illuminate gap junction connectivity, and pharmacologically perturb distinct subsets of cells. We expect this approach to have general utility for the specific delivery of many small molecules to defined cellular populations.