Filter
Associated Lab
- Aso Lab (30) Apply Aso Lab filter
- Betzig Lab (1) Apply Betzig Lab filter
- Bock Lab (2) Apply Bock Lab filter
- Branson Lab (9) Apply Branson Lab filter
- Card Lab (5) Apply Card Lab filter
- Clapham Lab (1) Apply Clapham Lab filter
- Dickson Lab (2) Apply Dickson Lab filter
- Druckmann Lab (1) Apply Druckmann Lab filter
- Fetter Lab (1) Apply Fetter Lab filter
- Funke Lab (2) Apply Funke Lab filter
- Harris Lab (3) Apply Harris Lab filter
- Heberlein Lab (2) Apply Heberlein Lab filter
- Hermundstad Lab (2) Apply Hermundstad Lab filter
- Hess Lab (6) Apply Hess Lab filter
- Jayaraman Lab (7) Apply Jayaraman Lab filter
- Lippincott-Schwartz Lab (1) Apply Lippincott-Schwartz Lab filter
- Looger Lab (2) Apply Looger Lab filter
- O'Shea Lab (1) Apply O'Shea Lab filter
- Otopalik Lab (1) Apply Otopalik Lab filter
- Reiser Lab (17) Apply Reiser Lab filter
- Riddiford Lab (1) Apply Riddiford Lab filter
- Romani Lab (2) Apply Romani Lab filter
- Rubin Lab (149) Apply Rubin Lab filter
- Saalfeld Lab (5) Apply Saalfeld Lab filter
- Scheffer Lab (8) Apply Scheffer Lab filter
- Schreiter Lab (1) Apply Schreiter Lab filter
- Simpson Lab (3) Apply Simpson Lab filter
- Singer Lab (1) Apply Singer Lab filter
- Spruston Lab (1) Apply Spruston Lab filter
- Stern Lab (1) Apply Stern Lab filter
- Svoboda Lab (3) Apply Svoboda Lab filter
- Tjian Lab (1) Apply Tjian Lab filter
- Truman Lab (4) Apply Truman Lab filter
- Turaga Lab (1) Apply Turaga Lab filter
- Turner Lab (5) Apply Turner Lab filter
- Zuker Lab (1) Apply Zuker Lab filter
Associated Project Team
- CellMap (1) Apply CellMap filter
- Fly Functional Connectome (4) Apply Fly Functional Connectome filter
- Fly Olympiad (3) Apply Fly Olympiad filter
- FlyEM (12) Apply FlyEM filter
- FlyLight (20) Apply FlyLight filter
- GENIE (1) Apply GENIE filter
- Transcription Imaging (1) Apply Transcription Imaging filter
Publication Date
- 2026 (2) Apply 2026 filter
- 2025 (8) Apply 2025 filter
- 2024 (4) Apply 2024 filter
- 2023 (5) Apply 2023 filter
- 2022 (1) Apply 2022 filter
- 2021 (4) Apply 2021 filter
- 2020 (9) Apply 2020 filter
- 2019 (6) Apply 2019 filter
- 2018 (7) Apply 2018 filter
- 2017 (15) Apply 2017 filter
- 2016 (3) Apply 2016 filter
- 2015 (16) Apply 2015 filter
- 2014 (9) Apply 2014 filter
- 2013 (5) Apply 2013 filter
- 2012 (8) Apply 2012 filter
- 2011 (4) Apply 2011 filter
- 2010 (4) Apply 2010 filter
- 2009 (2) Apply 2009 filter
- 2008 (4) Apply 2008 filter
- 2007 (2) Apply 2007 filter
- 2006 (1) Apply 2006 filter
- 2002 (1) Apply 2002 filter
- 2000 (2) Apply 2000 filter
- 1999 (1) Apply 1999 filter
- 1997 (1) Apply 1997 filter
- 1995 (2) Apply 1995 filter
- 1994 (2) Apply 1994 filter
- 1993 (2) Apply 1993 filter
- 1992 (1) Apply 1992 filter
- 1991 (2) Apply 1991 filter
- 1990 (3) Apply 1990 filter
- 1989 (2) Apply 1989 filter
- 1987 (2) Apply 1987 filter
- 1986 (1) Apply 1986 filter
- 1985 (1) Apply 1985 filter
- 1984 (1) Apply 1984 filter
- 1983 (1) Apply 1983 filter
- 1982 (2) Apply 1982 filter
- 1981 (1) Apply 1981 filter
- 1979 (1) Apply 1979 filter
- 1973 (1) Apply 1973 filter
Type of Publication
149 Publications
Showing 101-110 of 149 resultsWe have conducted a genetic screen for mutations that decrease the effectiveness of signaling by a protein tyrosine kinase, the product of the Drosophila melanogaster sevenless gene. These mutations define seven genes whose wild-type products may be required for signaling by sevenless. Four of the seven genes also appear to be essential for signaling by a second protein tyrosine kinase, the product of the Ellipse gene. The putative products of two of these seven genes have been identified. One encodes a ras protein. The other locus encodes a protein that is homologous to the S. cerevisiae CDC25 protein, an activator of guanine nucleotide exchange by ras proteins. These results suggest that the stimulation of ras protein activity is a key element in the signaling by sevenless and Ellipse and that this stimulation may be achieved by activating the exchange of GTP for bound GDP by the ras protein.
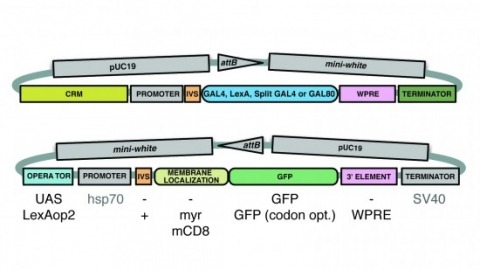
A wide variety of biological experiments rely on the ability to express an exogenous gene in a transgenic animal at a defined level and in a spatially and temporally controlled pattern. We describe major improvements of the methods available for achieving this objective in Drosophila melanogaster. We have systematically varied core promoters, UTRs, operator sequences, and transcriptional activating domains used to direct gene expression with the GAL4, LexA, and Split GAL4 transcription factors and the GAL80 transcriptional repressor. The use of site-specific integration allowed us to make quantitative comparisons between different constructs inserted at the same genomic location. We also characterized a set of PhiC31 integration sites for their ability to support transgene expression of both drivers and responders in the nervous system. The increased strength and reliability of these optimized reagents overcome many of the previous limitations of these methods and will facilitate genetic manipulations of greater complexity and sophistication.
Animals exhibit a behavioral response to novel sensory stimuli about which they have no prior knowledge. We have examined the neural and behavioral correlates of novelty and familiarity in the olfactory system of Drosophila. Novel odors elicit strong activity in output neurons (MBONs) of the α'3 compartment of the mushroom body that is rapidly suppressed upon repeated exposure to the same odor. This transition in neural activity upon familiarization requires odor-evoked activity in the dopaminergic neuron innervating this compartment. Moreover, exposure of a fly to novel odors evokes an alerting response that can also be elicited by optogenetic activation of α'3 MBONs. Silencing these MBONs eliminates the alerting behavior. These data suggest that the α'3 compartment plays a causal role in the behavioral response to novel and familiar stimuli as a consequence of dopamine-mediated plasticity at the Kenyon cell-MBONα'3 synapse.
Insecticides remain indispensable for crop protection and food security, yet their widespread use may contribute to the global decline of beneficial insect populations. Efforts to mitigate these impacts are hampered by a fragmented understanding of how insects metabolise insecticides and how sublethal exposures affect physiology, behaviour, and fitness. Here, we synthesise current understanding of metabolic detoxification and highlight critical gaps: the tissue- and time-dependent dynamics of insecticide entry and processing, the triggers and architecture of xenobiotic transcriptional responses, the role of rapid non-transcriptional regulation, and the population-level consequences of sublethal effects. We also outline emerging experimental strategies for addressing these questions and propose a next-generation research pipeline centred on multi-endpoint phenomics across life stages and sentinel species, integrated with AI-driven predictive toxicology, as a framework for identifying safer chemicals. We propose an integrated framework unifying molecular, physiological, and ecological responses to sublethal exposure to guide the design of insecticides that maintain effective pest control while safeguarding insect biodiversity and the ecosystems it underpins.
Dopamine signals reward in animal brains. A single presentation of a sugar reward to Drosophila activates distinct subsets of dopamine neurons that independently induce short- and long-term olfactory memories (STM and LTM, respectively). In this study, we show that a recurrent reward circuit underlies the formation and consolidation of LTM. This feedback circuit is composed of a single class of reward-signaling dopamine neurons (PAM-α1) projecting to a restricted region of the mushroom body (MB), and a specific MB output cell type, MBON-α1, whose dendrites arborize that same MB compartment. Both MBON-α1 and PAM-α1 neurons are required during the acquisition and consolidation of appetitive LTM. MBON-α1 additionally mediates the retrieval of LTM, which is dependent on the dopamine receptor signaling in the MB α/β neurons. Our results suggest that a reward signal transforms a nascent memory trace into a stable LTM using a feedback circuit at the cost of memory specificity.
The determination of cell fates during the assembly of the ommatidia in the compound eye of Drosophila appears to be controlled by cell-cell interactions. In this process, the sevenless gene is essential for the development of a single type of photoreceptor cell. In the absence of proper sevenless function the cells that would normally become the R7 photoreceptors instead become nonneuronal cells. Previous morphological and genetic analysis has indicated that the product of the sevenless gene is involved in reading or interpreting the positional information that specifies this particular developmental pathway. The sevenless gene has now been isolated and characterized. The data indicate that sevenless encodes a transmembrane protein with a tyrosine kinase domain. This structural similarity between sevenless and certain hormone receptors suggests that similar mechanisms are involved in developmental decisions based on cell-cell interaction and physiological or developmental changes induced by diffusible factors.
Sex differences in behaviour exist across the animal kingdom, typically under strong genetic regulation. In Drosophila, previous work has shown that fruitless and doublesex transcription factors identify neurons driving sexually dimorphic behaviour. However, the organisation of dimorphic neurons into functional circuits remains unclear.We now present the connectome of the entire Drosophila male central nervous system. This contains 166,691 neurons spanning the brain and ventral nerve cord, fully proofread and comprehensively annotated including fruitless and doublesex expression and 11,691 cell types. By comparison with a previous female brain connectome, we provide the first comprehensive description of the differences between male and female brains to synaptic resolution. Of 7,319 cross-matched cell types in the central brain, 114 are dimorphic with an additional 262 male- and 69 female-specific (totalling 4.8% of neurons in males and 2.4% in females).This resource enables analysis of full sensory-to-motor circuits underlying complex behaviours as well as the impact of dimorphic elements. Sex-specific and dimorphic neurons are concentrated in higher brain centres while the sensory and motor periphery are largely isomorphic. Within higher centres, male-specific connections are organised into hotspots defined by male-specific neurons or the presence of male-specific arbours on neurons that are otherwise similar between sexes. Numerous circuit switches reroute sensory information to form conserved, antagonistic circuits controlling opposing behaviours.
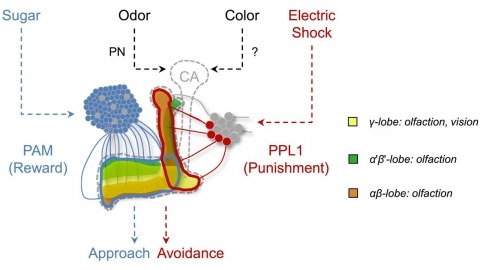
In nature, animals form memories associating reward or punishment with stimuli from different sensory modalities, such as smells and colors. It is unclear, however, how distinct sensory memories are processed in the brain. We established appetitive and aversive visual learning assays for Drosophila that are comparable to the widely used olfactory learning assays. These assays share critical features, such as reinforcing stimuli (sugar reward and electric shock punishment), and allow direct comparison of the cellular requirements for visual and olfactory memories. We found that the same subsets of dopamine neurons drive formation of both sensory memories. Furthermore, distinct yet partially overlapping subsets of mushroom body intrinsic neurons are required for visual and olfactory memories. Thus, our results suggest that distinct sensory memories are processed in a common brain center. Such centralization of related brain functions is an economical design that avoids the repetition of similar circuit motifs.
Animals rely on visual motion for navigating the world, and research in flies has clarified how neural circuits extract information from moving visual scenes. However, the major pathways connecting these patterns of optic flow to behavior remain poorly understood. Using a high-throughput quantitative assay of visually guided behaviors and genetic neuronal silencing, we discovered a region in Drosophila’s protocerebrum critical for visual motion following. We used neuronal silencing, calcium imaging, and optogenetics to identify a single cell type, LPC1, that innervates this region, detects translational optic flow, and plays a key role in regulating forward walking. Moreover, the population of LPC1s can estimate the travelling direction, such as when gaze direction diverges from body heading. By linking specific cell types and their visual computations to specific behaviors, our findings establish a foundation for understanding how the nervous system uses vision to guide navigation.
Animals are often bombarded with visual information and must prioritize specific visual features based on their current needs. The neuronal circuits that detect and relay visual features have been well studied. Much less is known about how an animal adjusts its visual attention as its goals or environmental conditions change. During social behaviours, flies need to focus on nearby flies. Here we study how the flow of visual information is altered when female Drosophila enter an aggressive state. From the connectome, we identify three state-dependent circuit motifs poised to modify the response of an aggressive female to fly-sized visual objects: convergence of excitatory inputs from neurons conveying select visual features and internal state; dendritic disinhibition of select visual feature detectors; and a switch that toggles between two visual feature detectors. Using cell-type-specific genetic tools, together with behavioural and neurophysiological analyses, we show that each of these circuit motifs is used during female aggression. We reveal that features of this same switch operate in male Drosophila during courtship pursuit, suggesting that disparate social behaviours may share circuit mechanisms. Our study provides a compelling example of using the connectome to infer circuit mechanisms that underlie dynamic processing of sensory signals.